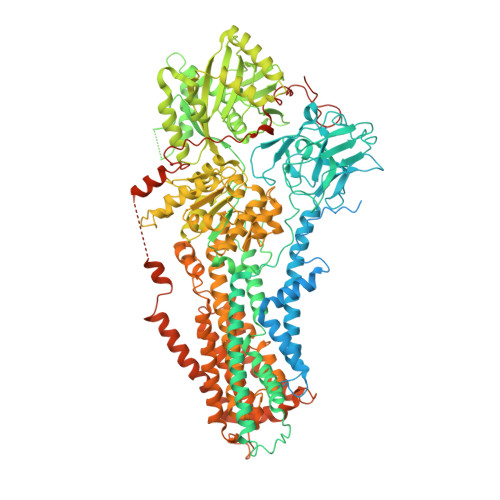
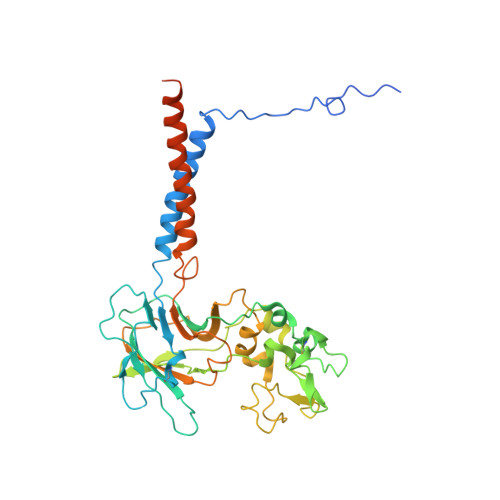
Autoinhibition and activation mechanisms of the eukaryotic lipid flippase Drs2p-Cdc50p.
Bai, L., Kovach, A., You, Q., Hsu, H.C., Zhao, G., Li, H.(2019) Nat Commun 10: 4142-4142
- PubMed: 31515475 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-019-12191-9
- Primary Citation Related Structures:
6PSX, 6PSY - PubMed Abstract:
The heterodimeric eukaryotic Drs2p-Cdc50p complex is a lipid flippase that maintains cell membrane asymmetry. The enzyme complex exists in an autoinhibited form in the absence of an activator and is specifically activated by phosphatidylinositol-4-phosphate (PI4P), although the underlying mechanisms have been unclear. Here we report the cryo-EM structures of intact Drs2p-Cdc50p isolated from S. cerevisiae in apo form and in the PI4P-activated form at 2.8 Å and 3.3 Å resolution, respectively. The structures reveal that the Drs2p C-terminus lines a long groove in the cytosolic regulatory region to inhibit the flippase activity. PIP4 binding in a cytosol-proximal membrane region triggers a 90° rotation of a cytosolic helix switch that is located just upstream of the inhibitory C-terminal peptide. The rotation of the helix switch dislodges the C-terminus from the regulatory region, activating the flippase.
- Structural Biology Program, Van Andel Research Institute, Grand Rapids, MI, 49503, USA. Lin.Bai@vai.org.
Organizational Affiliation: