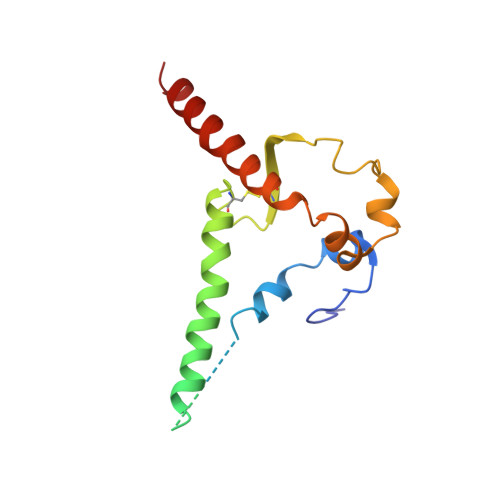
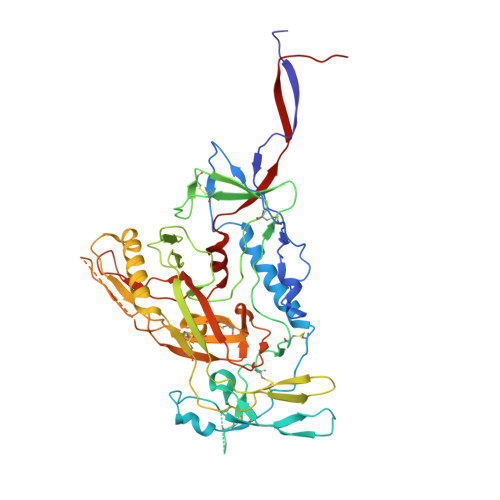
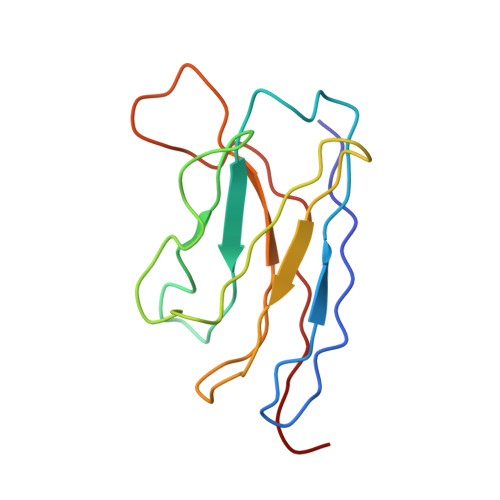
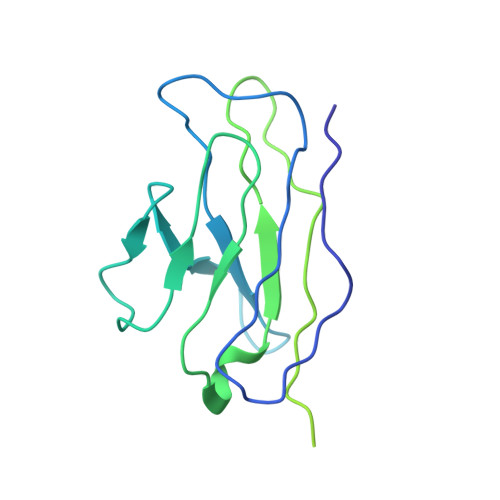
Immunization expands B cells specific to HIV-1 V3 glycan in mice and macaques.
Escolano, A., Gristick, H.B., Abernathy, M.E., Merkenschlager, J., Gautam, R., Oliveira, T.Y., Pai, J., West Jr., A.P., Barnes, C.O., Cohen, A.A., Wang, H., Golijanin, J., Yost, D., Keeffe, J.R., Wang, Z., Zhao, P., Yao, K.H., Bauer, J., Nogueira, L., Gao, H., Voll, A.V., Montefiori, D.C., Seaman, M.S., Gazumyan, A., Silva, M., McGuire, A.T., Stamatatos, L., Irvine, D.J., Wells, L., Martin, M.A., Bjorkman, P.J., Nussenzweig, M.C.(2019) Nature 570: 468-473
- PubMed: 31142836 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41586-019-1250-z
- Primary Citation Related Structures:
6ORN, 6ORO, 6ORP, 6ORQ - PubMed Abstract:
Broadly neutralizing monoclonal antibodies protect against infection with HIV-1 in animal models, suggesting that a vaccine that elicits these antibodies would be protective in humans. However, it has not yet been possible to induce adequate serological responses by vaccination. Here, to activate B cells that express precursors of broadly neutralizing antibodies within polyclonal repertoires, we developed an immunogen, RC1, that facilitates the recognition of the variable loop 3 (V3)-glycan patch on the envelope protein of HIV-1. RC1 conceals non-conserved immunodominant regions by the addition of glycans and/or multimerization on virus-like particles. Immunization of mice, rabbits and rhesus macaques with RC1 elicited serological responses that targeted the V3-glycan patch. Antibody cloning and cryo-electron microscopy structures of antibody-envelope complexes confirmed that immunization with RC1 expands clones of B cells that carry the anti-V3-glycan patch antibodies, which resemble precursors of human broadly neutralizing antibodies. Thus, RC1 may be a suitable priming immunogen for sequential vaccination strategies in the context of polyclonal repertoires.
- Laboratory of Molecular Immunology, The Rockefeller University, New York, NY, USA.
Organizational Affiliation: