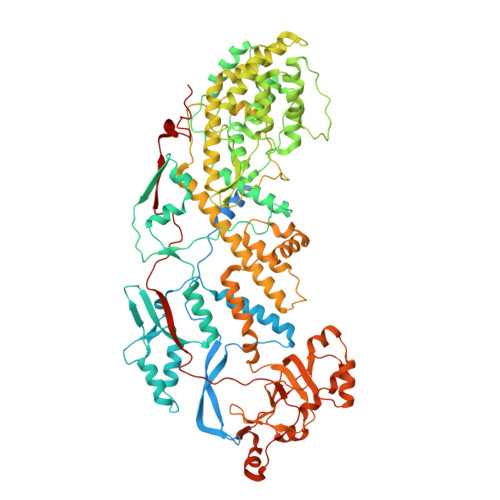
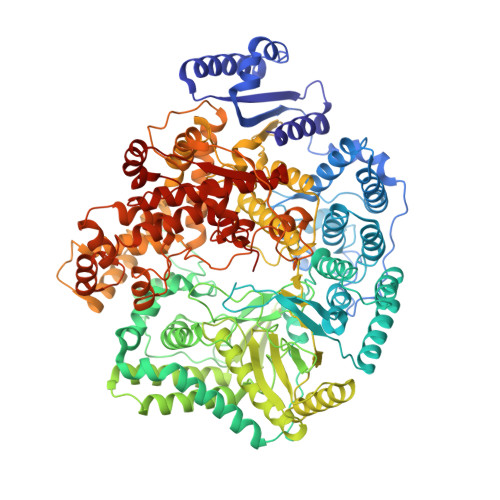
In situ Structure of Rotavirus VP1 RNA-Dependent RNA Polymerase.
Jenni, S., Salgado, E.N., Herrmann, T., Li, Z., Grant, T., Grigorieff, N., Trapani, S., Estrozi, L.F., Harrison, S.C.(2019) J Mol Biology 431: 3124-3138
- PubMed: 31233764
- DOI: https://doi.org/10.1016/j.jmb.2019.06.016
- Primary Citation Related Structures:
6OJ3, 6OJ4, 6OJ5, 6OJ6 - PubMed Abstract:
Rotaviruses, like other non-enveloped, double-strand RNA viruses, package an RNA-dependent RNA polymerase (RdRp) with each duplex of their segmented genomes. Rotavirus cell entry results in loss of an outer protein layer and delivery into the cytosol of an intact, inner capsid particle (the "double-layer particle," or DLP). The RdRp, designated VP1, is active inside the DLP; each VP1 achieves many rounds of mRNA transcription from its associated genome segment. Previous work has shown that one VP1 molecule lies close to each 5-fold axis of the icosahedrally symmetric DLP, just beneath the inner surface of its protein shell, embedded in tightly packed RNA. We have determined a high-resolution structure for the rotavirus VP1 RdRp in situ, by local reconstruction of density around individual 5-fold positions. We have analyzed intact virions ("triple-layer particles"), non-transcribing DLPs and transcribing DLPs. Outer layer dissociation enables the DLP to synthesize RNA, in vitro as well as in vivo, but appears not to induce any detectable structural change in the RdRp. Addition of NTPs, Mg 2+ , and S-adenosylmethionine, which allows active transcription, results in conformational rearrangements, in both VP1 and the DLP capsid shell protein, that allow a transcript to exit the polymerase and the particle. The position of VP1 (among the five symmetrically related alternatives) at one vertex does not correlate with its position at other vertices. This stochastic distribution of site occupancies limits long-range order in the 11-segment, double-strand RNA genome.
- Department of Biological Chemistry and Molecular Pharmacology, Harvard Medical School, 250 Longwood Avenue, Boston, MA 02115, USA.
Organizational Affiliation: