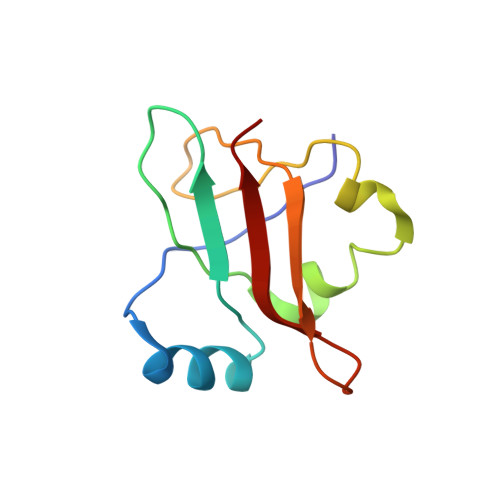
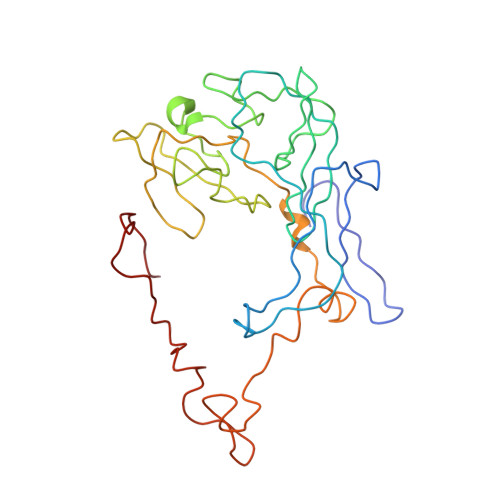
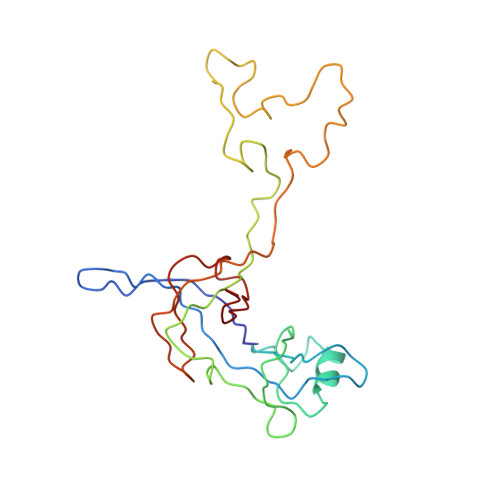
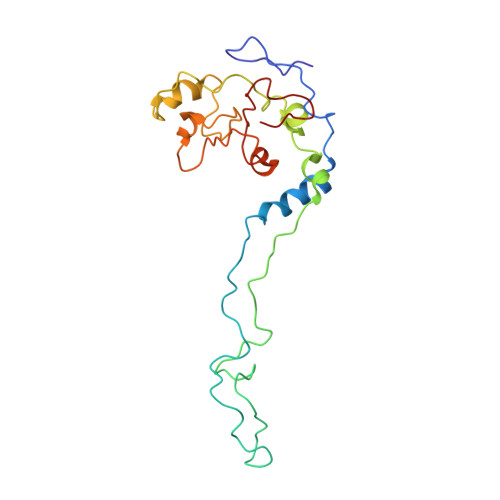
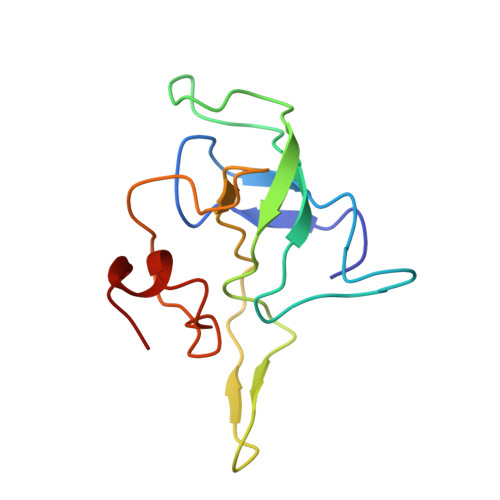
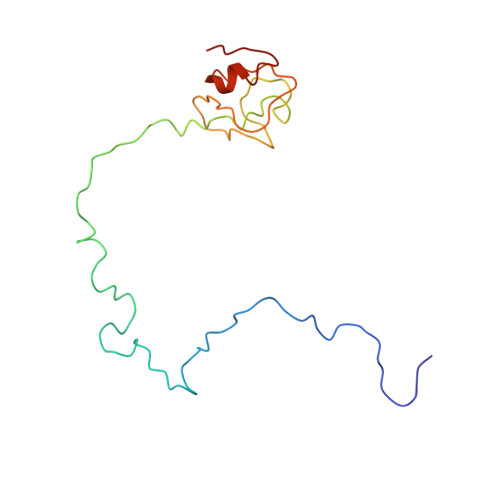
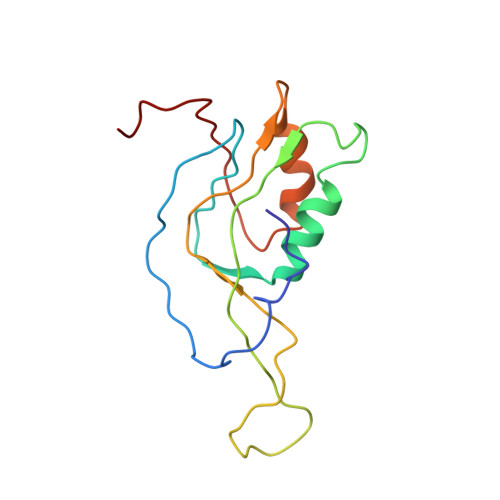
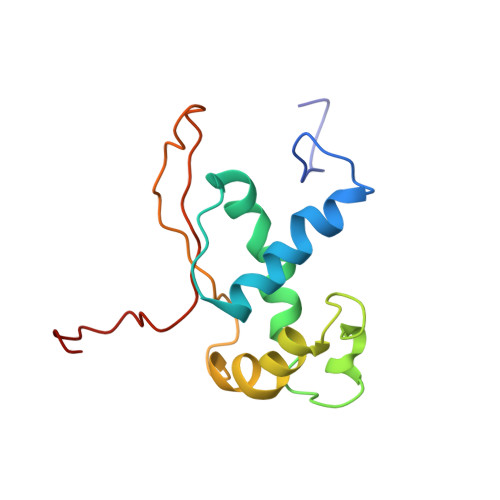
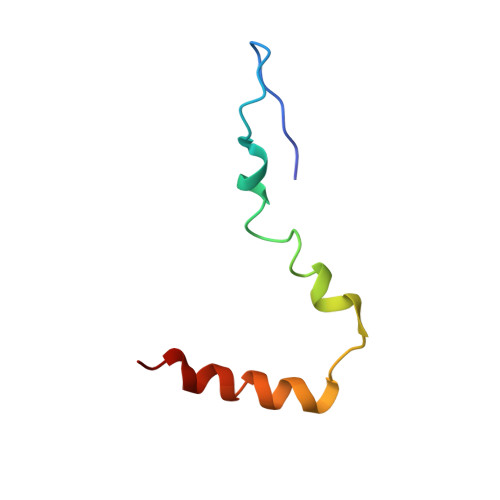
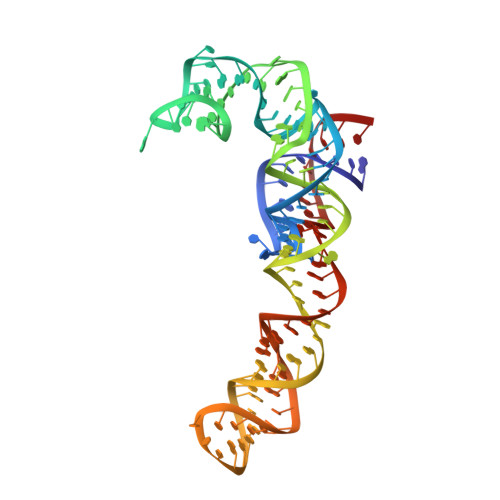
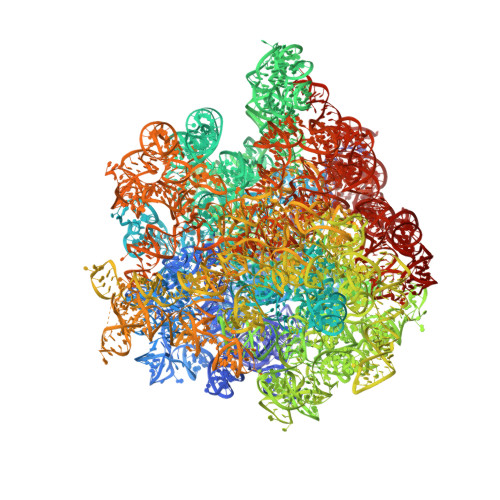
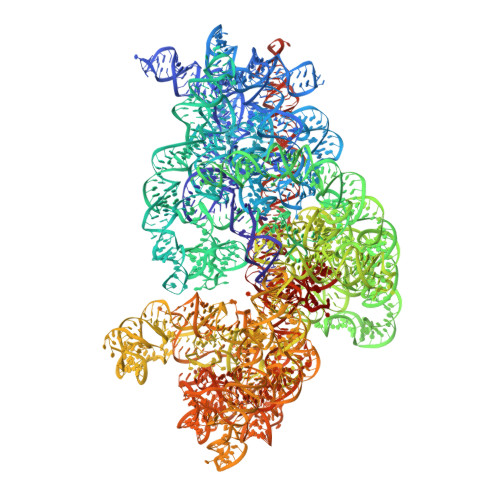
Late steps in bacterial translation initiation visualized using time-resolved cryo-EM.
Kaledhonkar, S., Fu, Z., Caban, K., Li, W., Chen, B., Sun, M., Gonzalez Jr., R.L., Frank, J.(2019) Nature 570: 400-404
- PubMed: 31108498 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41586-019-1249-5
- Primary Citation Related Structures:
6O7K, 6O9J, 6O9K - PubMed Abstract:
The initiation of bacterial translation involves the tightly regulated joining of the 50S ribosomal subunit to an initiator transfer RNA (fMet-tRNA fMet )-containing 30S ribosomal initiation complex to form a 70S initiation complex, which subsequently matures into a 70S elongation-competent complex. Rapid and accurate formation of the 70S initiation complex is promoted by initiation factors, which must dissociate from the 30S initiation complex before the resulting 70S elongation-competent complex can begin the elongation of translation 1 . Although comparisons of the structures of the 30S 2-5 and 70S 4,6-8 initiation complexes have revealed that the ribosome, initiation factors and fMet-tRNA fMet can acquire different conformations in these complexes, the timing of conformational changes during formation of the 70S initiation complex, the structures of any intermediates formed during these rearrangements, and the contributions that these dynamics might make to the mechanism and regulation of initiation remain unknown. Moreover, the absence of a structure of the 70S elongation-competent complex formed via an initiation-factor-catalysed reaction has precluded an understanding of the rearrangements to the ribosome, initiation factors and fMet-tRNA fMet that occur during maturation of a 70S initiation complex into a 70S elongation-competent complex. Here, using time-resolved cryogenic electron microscopy 9 , we report the near-atomic-resolution view of how a time-ordered series of conformational changes drive and regulate subunit joining, initiation factor dissociation and fMet-tRNA fMet positioning during formation of the 70S elongation-competent complex. Our results demonstrate the power of time-resolved cryogenic electron microscopy to determine how a time-ordered series of conformational changes contribute to the mechanism and regulation of one of the most fundamental processes in biology.
- Department of Biochemistry & Molecular Biophysics, Columbia University, New York, NY, USA.
Organizational Affiliation: