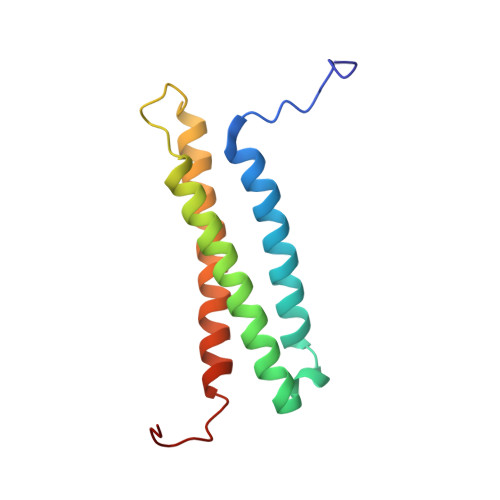
The N-Terminal Domain of NPHP1 Folds into a Monomeric Left-Handed Antiparallel Three-Stranded Coiled Coil with Anti-apoptotic Function.
Mannella, V., Quilici, G., Nigro, E.A., Lampis, M., Minici, C., Degano, M., Boletta, A., Musco, G.(2019) ACS Chem Biol 14: 1845-1854
- PubMed: 31345020
- DOI: https://doi.org/10.1021/acschembio.9b00582
- Primary Citation of Related Structures:
6O1Q - PubMed Abstract:
Mutations in the NPHP1 gene, coding for human nephrocystin-1 (NPHP1), cause the autosomal recessive disease nephronophthisis, the most common cause of end-stage renal disease in children and adolescents. The function and structure of NPHP1 are still poorly characterized. NPHP1 presents a modular structure well in keeping with its role as an adaptor protein: it harbors an SH3 domain flanked by two glutamic acid-rich regions and a conserved C-terminal nephrocystin homology domain (NHD). Similar to other NPHP protein family members, its N-terminus contains a putative coiled-coil domain (NPHP1 CC ) that is supposed to play an important role in NPHP1 self-association and/or protein-protein interactions. Structural studies proving its structure and its oligomerization state are still lacking. Here we demonstrate that NPHP1 CC is monomeric in solution and unexpectedly folds into an autonomous domain forming a three-stranded antiparallel coiled coil suitable for protein-protein interactions. Notably, we found that the NPHP1 CC shares remarkable structural similarities with the three-stranded coiled coil of the BAG domain protein family, which is known to mediate the anti-apoptotic function of these proteins, suggesting a possible similar role for NPHP1 CC . In agreement with this hypothesis, we show that in the context of the full-length protein the NPHP1 CC is fundamental to regulate resistance to apoptotic stimuli.
- Biomolecular NMR Laboratory, Division of Genetics and Cell Biology , IRCCS Ospedale San Raffaele , Via Olgettina 58 , 20132 Milan , Italy.
Organizational Affiliation: