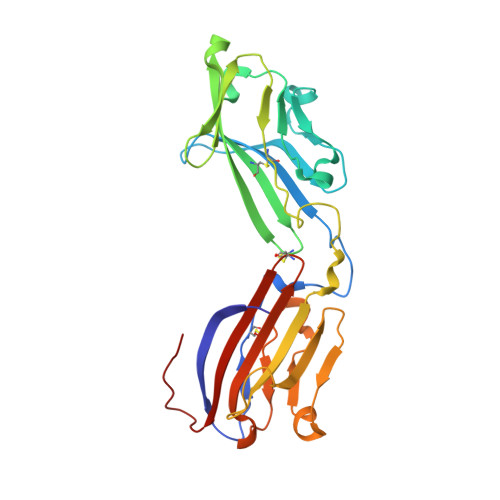
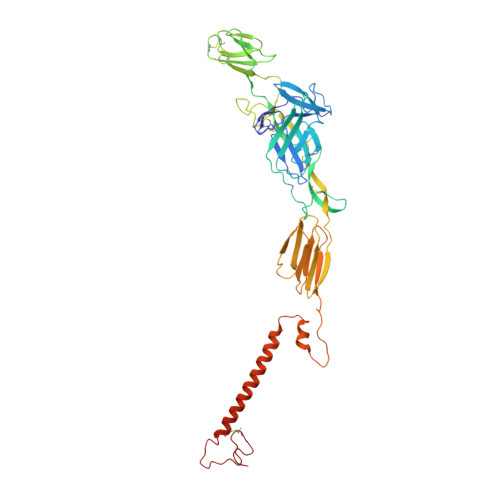
Cryo-EM Structure of Chikungunya Virus in Complex with the Mxra8 Receptor.
Basore, K., Kim, A.S., Nelson, C.A., Zhang, R., Smith, B.K., Uranga, C., Vang, L., Cheng, M., Gross, M.L., Smith, J., Diamond, M.S., Fremont, D.H.(2019) Cell 177: 1725
- PubMed: 31080061 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.cell.2019.04.006
- Primary Citation Related Structures:
6NK3, 6NK5, 6NK6, 6NK7 - PubMed Abstract:
Mxra8 is a receptor for multiple arthritogenic alphaviruses that cause debilitating acute and chronic musculoskeletal disease in humans. Herein, we present a 2.2 Å resolution X-ray crystal structure of Mxra8 and 4 to 5 Å resolution cryo-electron microscopy reconstructions of Mxra8 bound to chikungunya (CHIKV) virus-like particles and infectious virus. The Mxra8 ectodomain contains two strand-swapped Ig-like domains oriented in a unique disulfide-linked head-to-head arrangement. Mxra8 binds by wedging into a cleft created by two adjacent CHIKV E2-E1 heterodimers in one trimeric spike and engaging a neighboring spike. Two binding modes are observed with the fully mature VLP, with one Mxra8 binding with unique contacts. Only the high-affinity binding mode was observed in the complex with infectious CHIKV, as viral maturation and E3 occupancy appear to influence receptor binding-site usage. Our studies provide insight into how Mxra8 binds CHIKV and creates a path for developing alphavirus entry inhibitors.
- Department of Pathology & Immunology, Washington University School of Medicine, Saint Louis, MO 63110, USA.
Organizational Affiliation: