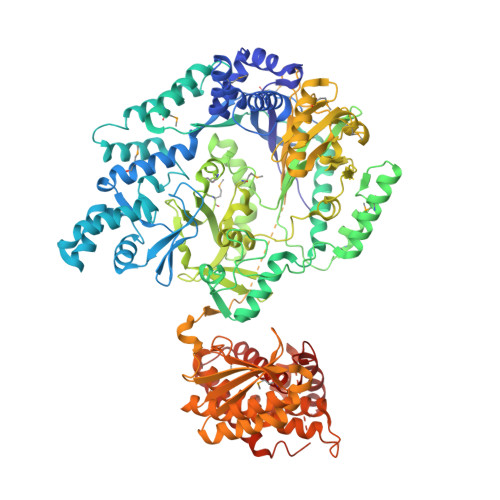
Characterization of glutamyl-tRNA-dependent dehydratases using nonreactive substrate mimics.
Bothwell, I.R., Cogan, D.P., Kim, T., Reinhardt, C.J., van der Donk, W.A., Nair, S.K.(2019) Proc Natl Acad Sci U S A 116: 17245-17250
- PubMed: 31409709 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1905240116
- Primary Citation Related Structures:
6EC7, 6EC8, 6M7Y - PubMed Abstract:
The peptide natural product nisin has been used as a food preservative for 6 decades with minimal development of resistance. Nisin contains the unusual amino acids dehydroalanine and dehydrobutyrine, which are posttranslationally installed by class I lanthipeptide dehydratases (LanBs) on a linear peptide substrate through an unusual glutamyl-tRNA-dependent dehydration of Ser and Thr. To date, little is known about how LanBs catalyze the transfer of glutamate from charged tRNA Glu to the peptide substrate, or how they carry out the subsequent elimination of the peptide-glutamyl adducts to afford dehydro amino acids. Here, we describe the synthesis of inert analogs that mimic substrate glutamyl-tRNA Glu and the glutamylated peptide intermediate, and determine the crystal structures of 2 LanBs in complex with each of these compounds. Mutational studies were used to characterize the function of the glutamylation and glutamate elimination active-site residues identified through the structural analysis. These combined studies provide insights into the mechanisms of substrate recognition, glutamylation, and glutamate elimination by LanBs to effect a net dehydration reaction of Ser and Thr.
- Department of Chemistry, University of Illinois at Urbana-Champaign, Urbana, IL 61801.
Organizational Affiliation: