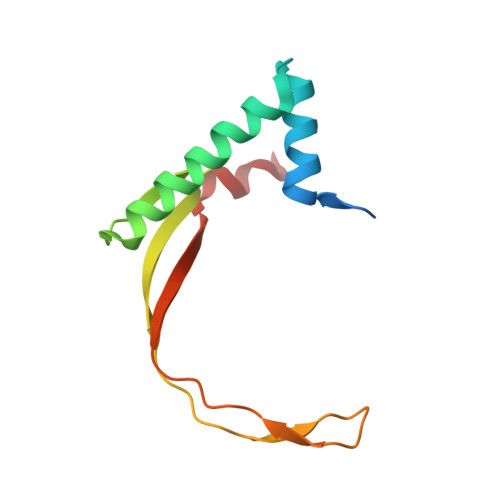
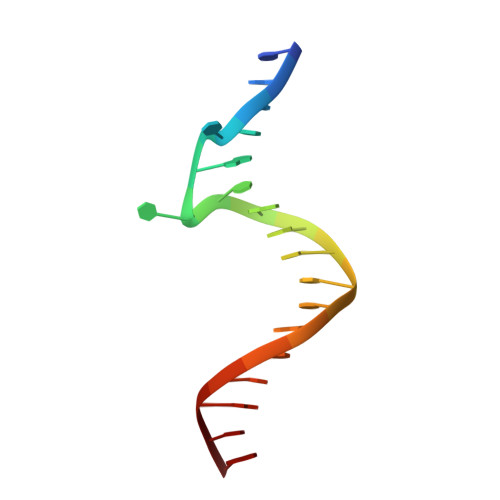
The structural basis of African swine fever virus pA104R binding to DNA and its inhibition by stilbene derivatives.
Liu, R., Sun, Y., Chai, Y., Li, S., Li, S., Wang, L., Su, J., Yu, S., Yan, J., Gao, F., Zhang, G., Qiu, H.J., Gao, G.F., Qi, J., Wang, H.(2020) Proc Natl Acad Sci U S A 117: 11000-11009
- PubMed: 32358196 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1922523117
- Primary Citation Related Structures:
6LMH, 6LMJ - PubMed Abstract:
African swine fever virus (ASFV) is a highly contagious nucleocytoplasmic large DNA virus (NCLDV) that causes nearly 100% mortality in swine. The development of effective vaccines and drugs against this virus is urgently needed. pA104R, an ASFV-derived histone-like protein, shares sequence and functional similarity with bacterial HU/IHF family members and is essential for viral replication. Herein, we solved the crystal structures of pA104R in its apo state as well as in complex with DNA. Apo-pA104R forms a homodimer and folds into an architecture conserved in bacterial heat-unstable nucleoid proteins/integration host factors (HUs/IHFs). The pA104R-DNA complex structure, however, uncovers that pA104R has a DNA binding pattern distinct from its bacterial homologs, that is, the β-ribbon arms of pA104R stabilize DNA binding by contacting the major groove instead of the minor groove. Mutations of the basic residues at the base region of the β-strand DNA binding region (BDR), rather than those in the β-ribbon arms, completely abolished DNA binding, highlighting the major role of the BDR base in DNA binding. An overall DNA bending angle of 93.8° is observed in crystal packing of the pA104R-DNA complex structure, which is close to the DNA bending angle in the HU-DNA complex. Stilbene derivatives SD1 and SD4 were shown to disrupt the binding between pA104R and DNA and inhibit the replication of ASFV in primary porcine alveolar macrophages. Collectively, these results reveal the structural basis of pA104R binding to DNA highlighting the importance of the pA104R-DNA interaction in the ASFV replication cycle and provide inhibitor leads for ASFV chemotherapy.
- College of Animal Science and Veterinary Medicine, Henan Agricultural University, 450046 Zhengzhou, Henan, China.
Organizational Affiliation: