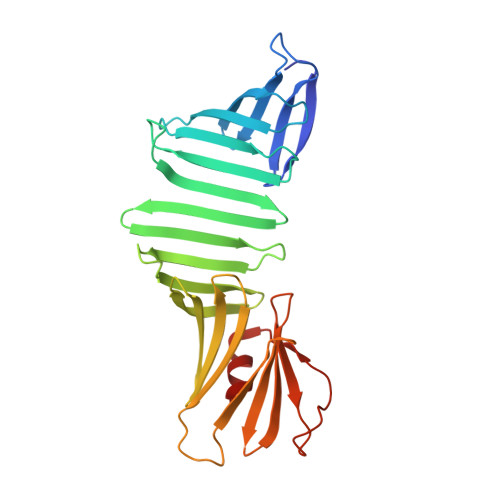
beta-Strand-mediated Domain-swapping in the Absence of Hydrophobic Core Repacking.
Kiya, M., Shiga, S., Ding, P., Koide, S., Makabe, K.(2024) J Mol Biology 436: 168405-168405
- PubMed: 38104859 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2023.168405
- Primary Citation Related Structures:
6KWJ, 6KWU, 6KWV, 6LJY, 7FDD, 7FJR - PubMed Abstract:
Domain swapping is a process wherein a portion of a protein is exchanged with its counterpart in another copy of the molecule, resulting in the formation of homo-oligomers with concomitant repacking of a hydrophobic core. Here, we report domain swapping triggered upon modifying a β-hairpin sequence within a single-layer β-sheet (SLB) of a model protein, OspA that did not involve the formation of a reorganized hydrophobic core. The replacement of two β-hairpin sequences with a Gly-Gly and shorteing of a β-hairpin resulted in a protein that formed two distinct crystal structures under similar conditions: one was monomeric, similar to the parental molecule, whereas the other was a domain-swapped dimer, mediated by an intermolecular β-sheet in the SLB portion. Based on the dimer interface structure, we replaced the Gly-Gly sequence with three-residue sequences that enable the formation of a consecutive intermolecular β-sheet, including the Cys-Thr-Cys sequence that formed a stable disulfide-linked dimer. These results provide new insights into protein folding, evolution, and the designability of protein structure.
- Graduate School of Science and Engineering, Yamagata University, 4-3-16 Jyonan, Yonezawa, Yamagata 992-8510, Japan.
Organizational Affiliation: