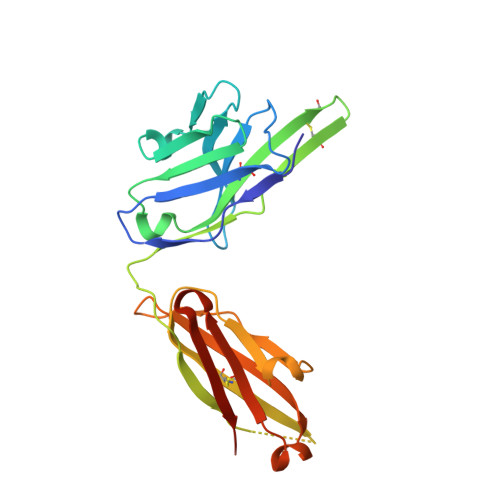
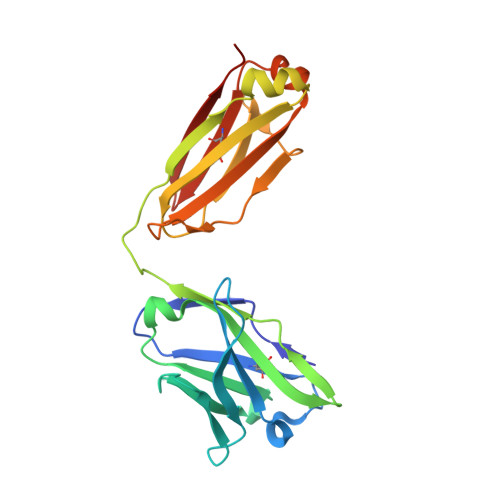
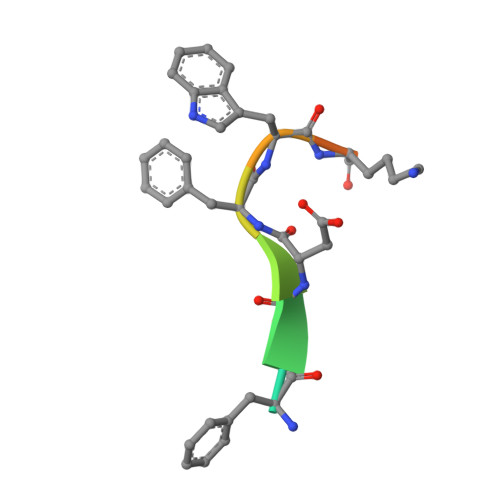
Selection of a picomolar antibody that targets CXCR2-mediated neutrophil activation and alleviates EAE symptoms.
Shi, X., Wan, Y., Wang, N., Xiang, J., Wang, T., Yang, X., Wang, J., Dong, X., Dong, L., Yan, L., Li, Y., Liu, L., Hou, S., Zhong, Z., Wilson, I.A., Yang, B., Yang, G., Lerner, R.A.(2021) Nat Commun 12: 2547-2547
- PubMed: 33953162 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-021-22810-z
- Primary Citation Related Structures:
6KVA, 6KVF - PubMed Abstract:
Receptors and their ligands are important therapeutic targets for about one third of marketed drugs. Here, we describe an epitope-guided approach for selection of antibodies that modulate cellular signaling of targeted receptors. We chose CXC chemokine receptor 2 (CXCR2) in the G-protein coupled receptor superfamily as receptor and a CXCR2 N-terminal peptide for antibody selection. We obtain a highly selective, tight-binding antibody from a 10 11 -member antibody library using combinatorial enrichment. Structural and Hydrogen-Deuterium-Exchange mass spectrometry analyses demonstrate antibody interaction with an N-terminal region of CXCR2 that is part of the IL-8 epitope. The antibody strongly inhibits IL-8-induced and CXCR2-mediated neutrophil chemotaxis in vitro and alleviates hCXCR2-dependent experimental autoimmune encephalomyelitis symptoms in mice. As inappropriate neutrophil migration accompanies many diseases including inflammatory bowel disease, glomerulonephritis, allergic asthma, chronic obstructive pulmonary disease, and cancer, this antibody has potential for development as a therapeutic agent, akin to anti-TNF antibodies. However, an important difference here is that the antibody targets the chemokine receptor and competes with natural ligand, rather than targeting the ligand itself.
- Shanghai Institute for Advanced Immunochemical Studies, ShanghaiTech University, Shanghai, China.
Organizational Affiliation: