Structural insights into the substrate specificity of SP_0149, the substrate-binding protein of a methionine ABC transporter from Streptococcus pneumoniae.
Jha, B., Vyas, R., Bhushan, J., Sehgal, D., Biswal, B.K.(2019) Acta Crystallogr F Struct Biol Commun 75: 520-528
- PubMed: 31282873
- DOI: https://doi.org/10.1107/S2053230X19009038
- Primary Citation Related Structures:
6JF1 - PubMed Abstract:
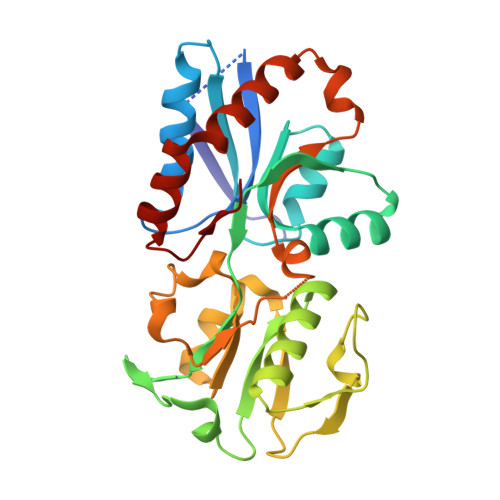
Successful pathogenesis is a cumulative effect of the virulence factors of a pathogen and its capability to efficiently utilize the available nutrients from the host. Streptococcus pneumoniae, a Gram-positive opportunistic pathogen, may either reside asymptomatically as a nasopharyngeal commensal inside the human host or cause lethal diseases, including pneumonia, meningitis and sepsis. S. pneumoniae is known to acquire methionine (Met) from its host through a Met importer. Here, the crystal structure of the substrate-binding protein (SBP; SP_0149) of an ABC importer with Met bound is reported at a resolution of 1.95 Å. The three-dimensional structure of SBP shows that it is composed of two distinct domains, each consisting of a mixed β-sheet flanked by helices. The substrate, Met, is bound in the central part of the interface between the two domains. The overall structure of SP_0149 resembles those of SBPs from other reported bacterial Met and Gly-Met dipeptide transporters. However, a detailed analysis of these structures shows notable variations in the amino-acid composition of the substrate-binding pockets of the SP_0149-Met and GmpC-Gly-Met structures. In particular, SP_0149 harbors Thr212 and Tyr114, whereas the corresponding residues in GmpC are Gly and Val. This difference is likely to be the underlying basis for their differential substrate specificity. In summary, the structure of the SP_0149-Met complex provides insights into the transport function of SP_0149 and its interactions with methionine. It opens up avenues for the rational design of inhibitors of SP_0149 through a structure-mediated approach.
- Structural and Functional Biology Laboratory, National Institute of Immunology, Aruna Asaf Ali Marg, New Delhi 110 067, India.
Organizational Affiliation: