Characterization of an SSB-dT25 complex: structural insights into the S-shaped ssDNA binding conformation.
Huang, Y.H., Chen, I.C., Huang, C.Y.(2019) RSC Adv 9: 40388-40396
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
(2019) RSC Adv 9: 40388-40396
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Single-stranded DNA-binding protein | 121 | Pseudomonas aeruginosa PAO1 | Mutation(s): 0 Gene Names: ssb, PA4232 | 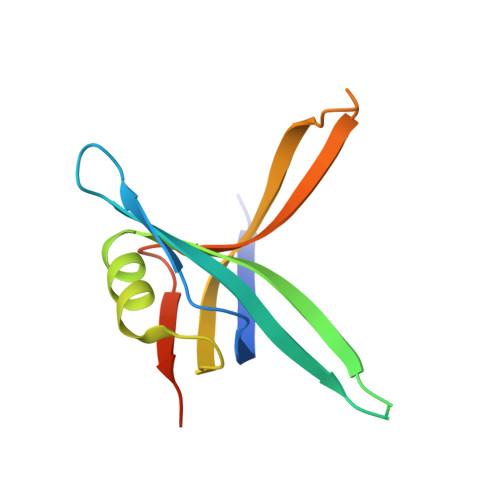 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P40947 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| DNA (25-MER) | 25 | synthetic construct |  | |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 60.488 | α = 90 |
| b = 60.488 | β = 90 |
| c = 131.315 | γ = 120 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| MOLREP | phasing |