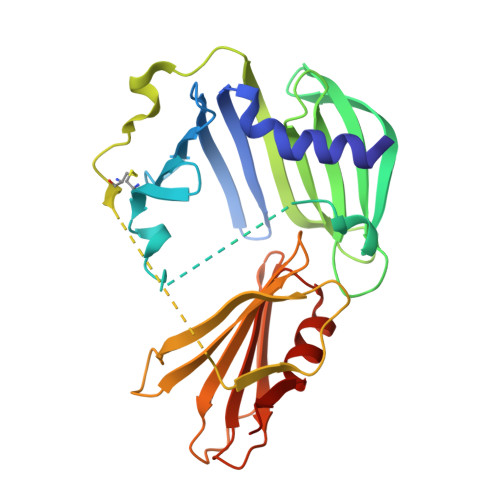
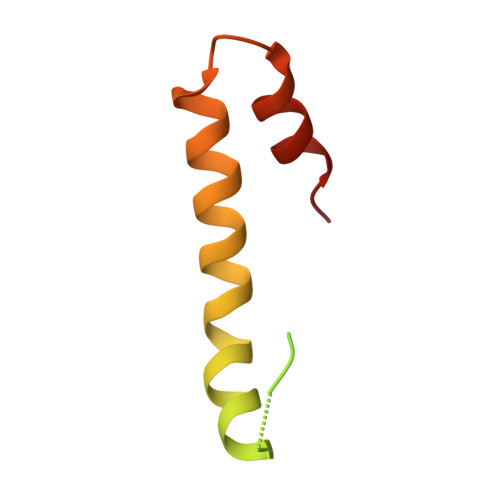
Structural basis for the recognition of MucA by MucB and AlgU in Pseudomonas aeruginosa.
Li, S., Lou, X., Xu, Y., Teng, X., Liu, R., Zhang, Q., Wu, W., Wang, Y., Bartlam, M.(2019) FEBS J 286: 4982-4994
- PubMed: 31297938 Search on PubMed
- DOI: https://doi.org/10.1111/febs.14995
- Primary Citation Related Structures:
6IN7, 6IN8, 6IN9 - PubMed Abstract:
Alginate production in Pseudomonas aeruginosa is regulated by the alternate σ factor AlgU, which in turn is regulated by the MucABCD system. The anti-σ factor MucA binds AlgU in the cytoplasm and prevents AlgU from binding to the RNA polymerase for transcription. MucB binds MucA in the periplasm and inhibits proteolysis of MucA and subsequent release of AlgU. In this work, we report crystal structures of MucA in complex with AlgU and MucB. A structure of MucB alone reveals the structural changes required for MucA recognition. A unique disulfide bond is identified in MucB, and mutation of this disulfide bond results in a shift from monomer to MucB dimers or tetramers. As MucB tetramers have previously been shown to be unable to bind MucA, this suggests a redox-sensitive stress response mechanism in MucB. The AlgU-MucA structure reveals a conserved σ factor/anti-σ factor complex, but AlgU lacks a disulfide bond conserved in many other σ factors. Our structures reveal the molecular basis for MucA recognition by MucB in the periplasm and AlgU in the cytoplasm, thus providing an important step in understanding the mechanisms that regulate a key signal transduction pathway involved in P. aeruginosa pathogenesis. DATABASE: The atomic coordinates and structure factors for MucA cyto -AlgU, MucB, and MucA peri -MucB have been deposited in the Protein Data Bank (PDB) with the accession code 6IN7, 6IN8, and 6IN9, respectively.
- State Key Laboratory of Medicinal Chemical Biology, Nankai University, Tianjin, China.
Organizational Affiliation: