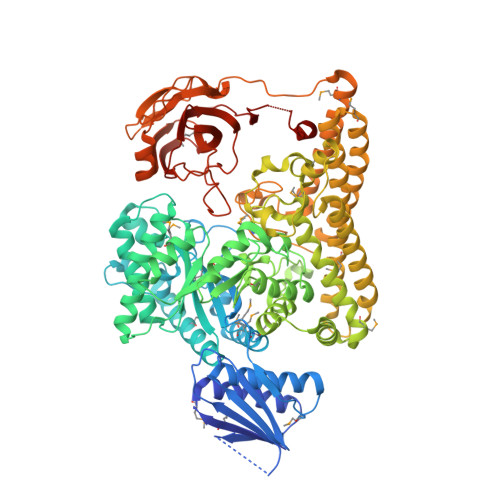
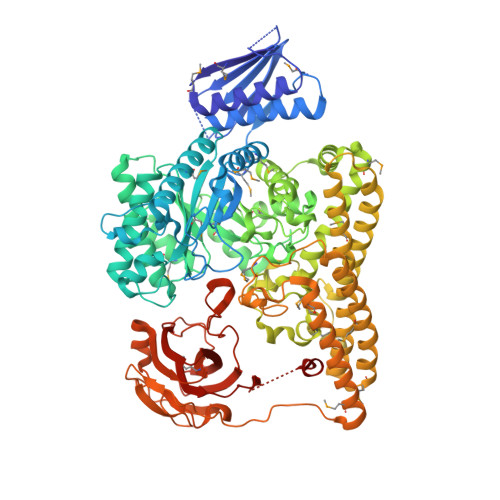
Structural and functional analyses of glycoside hydrolase 138 enzymes targeting chain A galacturonic acid in the complex pectin rhamnogalacturonan II.
Labourel, A., Basle, A., Munoz-Munoz, J., Ndeh, D., Booth, S., Nepogodiev, S.A., Field, R.A., Cartmell, A.(2019) J Biological Chem 294: 7711-7721
- PubMed: 30877196 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.RA118.006626
- Primary Citation Related Structures:
6HZE, 6HZF, 6HZG - PubMed Abstract:
The metabolism of carbohydrate polymers drives microbial diversity in the human gut microbiome. The selection pressures in this environment have spurred the evolution of a complex reservoir of microbial genes encoding carbohydrate-active enzymes (CAZymes). Previously, we have shown that the human gut bacterium Bacteroides thetaiotaomicron ( Bt ) can depolymerize the most structurally complex glycan, the plant pectin rhamnogalacturonan II (RGII), commonly found in the human diet. Previous investigation of the RGII-degrading apparatus in Bt identified BT0997 as a new CAZyme family, classified as glycoside hydrolase 138 (GH138). The mechanism of substrate recognition by GH138, however, remains unclear. Here, using synthetic substrates and biochemical assays, we show that BT0997 targets the d-galacturonic acid-α-1,2-l-rhamnose linkage in chain A of RGII and that it absolutely requires the presence of a second d-galacturonic acid side chain (linked β-1,3 to l-rhamnose) for activity. NMR analysis revealed that BT0997 operates through a double displacement retaining mechanism. We also report the crystal structure of a BT0997 homolog, BPA0997 from Bacteroides paurosaccharolyticus , in complex with ligands at 1.6 Å resolution. The structure disclosed that the enzyme comprises four domains, including a catalytic TIM (α/β) 8 barrel. Characterization of several BT0997 variants identified Glu-294 and Glu-361 as the catalytic acid/base and nucleophile, respectively, and we observed a chloride ion close to the active site. The three-dimensional structure and bioinformatic analysis revealed that two arginines, Arg-332 and Arg-521, are key specificity determinants of BT0997 in targeting d-galacturonic acid residues. In summary, our study reports the first structural and mechanistic analyses of GH138 enzymes.
- From the Institute for Cell and Molecular Biosciences, Newcastle University, Newcastle upon Tyne NE2 4HH, United Kingdom and.
Organizational Affiliation: