The structure of Proteus mirabilis N-acetylneuraminate lyase reveals an intermolecular disulphide bond
North, R.A., Garcia-Bonete, M.J., Goyal, P., Katona, G., Dobson, R.C.J., Friemann, R.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Putative N-acetylneuraminate lyase | 295 | Proteus mirabilis HI4320 | Mutation(s): 0 Gene Names: PMI2973 | 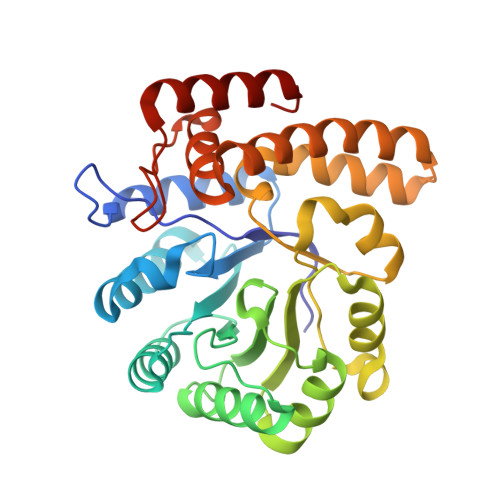 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | B4EZY4 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| SO4 Download:Ideal Coordinates CCD File | C [auth A], D [auth B] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 80.777 | α = 90 |
| b = 81.984 | β = 90 |
| c = 106.839 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |
| SCALA | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Royal Society of New Zealand | New Zealand | UOC1506 |
| Swedish Research Council | Sweden | 2011-5790 |
| Swedish Research Council | Sweden | 2010-1759 |
| European Molecular Biology Organization | New Zealand | 584-2014 |