Molecular mechanisms behind DAPK regulation: how phosphorylation switches work
Huart, A.-S., Simon, B., Lubner, J., Mertens, H.D.T., Temmerman, K., Hoffmann, J.-E., Svergun, D.I., Schwartz, D., Schultz, C., Wilmanns, M.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Death-associated protein kinase 1 | 337 | Homo sapiens | Mutation(s): 2 Gene Names: DAPK1, DAPK EC: 2.7.11.1 | 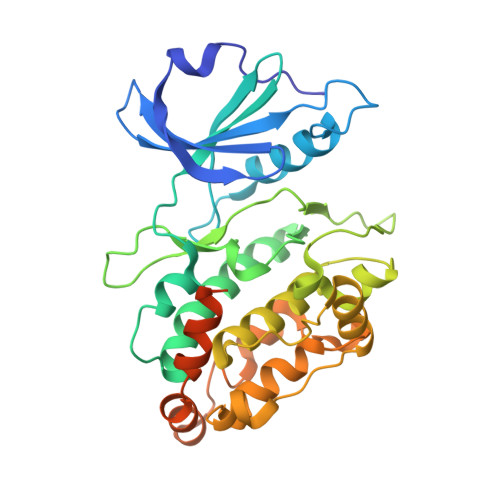 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P53355 GTEx: ENSG00000196730 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P53355 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 5 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| PEG Download:Ideal Coordinates CCD File | I [auth A] | DI(HYDROXYETHYL)ETHER C4 H10 O3 MTHSVFCYNBDYFN-UHFFFAOYSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | J [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| ACT Download:Ideal Coordinates CCD File | G [auth A] | ACETATE ION C2 H3 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-M |  | ||
| CL Download:Ideal Coordinates CCD File | B [auth A], C [auth A], D [auth A], E [auth A], F [auth A] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| MG Download:Ideal Coordinates CCD File | H [auth A] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 49.507 | α = 90 |
| b = 76.653 | β = 90 |
| c = 108.009 | γ = 90 |
| Software Name | Purpose |
|---|---|
| MxCuBE | data collection |
| XDS | data reduction |
| Aimless | data scaling |
| Coot | model building |
| PHENIX | refinement |
| PHASER | phasing |