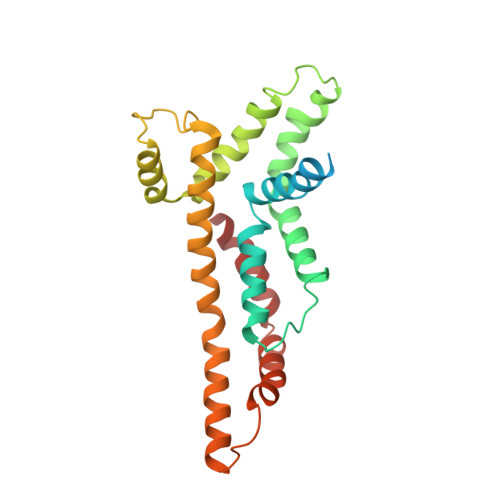
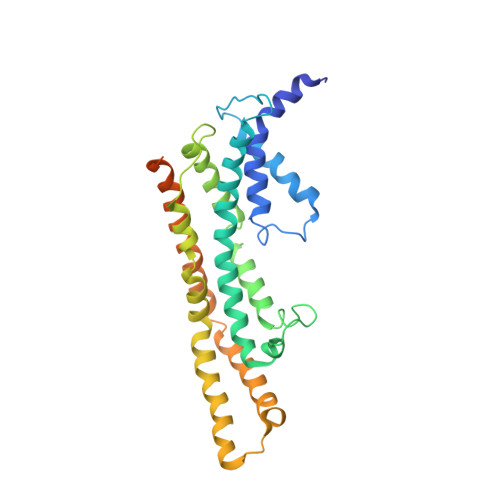
Structure of the core of the type III secretion system export apparatus.
Kuhlen, L., Abrusci, P., Johnson, S., Gault, J., Deme, J., Caesar, J., Dietsche, T., Mebrhatu, M.T., Ganief, T., Macek, B., Wagner, S., Robinson, C.V., Lea, S.M.(2018) Nat Struct Mol Biol 25: 583-590
- PubMed: 29967543 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41594-018-0086-9
- Primary Citation Related Structures:
6F2D - PubMed Abstract:
Export of proteins through type III secretion systems is critical for motility and virulence of many major bacterial pathogens. Three putative integral membrane proteins (FliP, FliQ, FliR) are suggested to form the core of an export gate in the inner membrane, but their structure, assembly and location within the final nanomachine remain unclear. Here, we present the cryoelectron microscopy structure of the Salmonella Typhimurium FliP-FliQ-FliR complex at 4.2 Å. None of the subunits adopt canonical integral membrane protein topologies, and common helix-turn-helix structural elements allow them to form a helical assembly with 5:4:1 stoichiometry. Fitting of the structure into reconstructions of intact secretion systems, combined with cross-linking, localize the export gate as a core component of the periplasmic portion of the machinery. This study thereby identifies the export gate as a key element of the secretion channel and implies that it primes the helical architecture of the components assembling downstream.
- Sir William Dunn School of Pathology, University of Oxford, Oxford, UK.
Organizational Affiliation: