Structural elements of a pH-sensitive inhibitor binding site in NMDA receptors
Regan, M.C., Furukawa, H.(2019) Nat Commun 10: 321
Experimental Data Snapshot
Starting Model: experimental
View more details
(2019) Nat Commun 10: 321
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Glutamate receptor ionotropic, NMDA 1 | 385 | Xenopus laevis | Mutation(s): 2 Gene Names: grin1 Membrane Entity: Yes | 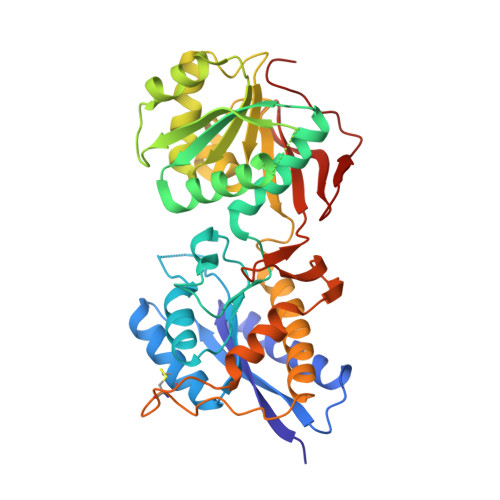 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A1L8F5J9 | ||||
Glycosylation | |||||
| Glycosylation Sites: 2 | |||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Glutamate receptor ionotropic, NMDA 2B | 363 | Rattus norvegicus | Mutation(s): 1 Gene Names: Grin2b Membrane Entity: Yes | 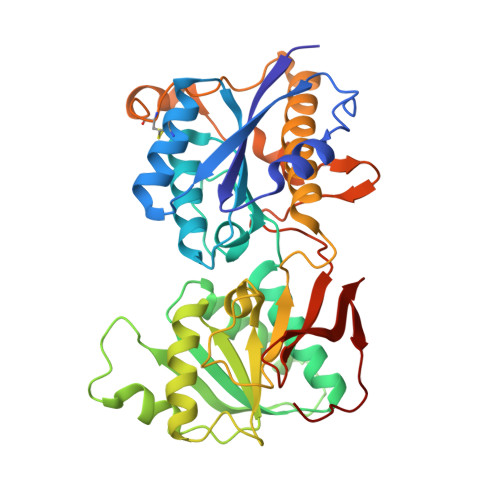 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q00960 | ||||
Glycosylation | |||||
| Glycosylation Sites: 2 | Go to GlyGen: Q00960-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | E | 5 |  | N-Glycosylation | |
Glycosylation Resources | |||||
GlyTouCan: G22768VO GlyCosmos: G22768VO GlyGen: G22768VO | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| T97 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | L [auth B], V [auth D] | N-{4-[(2S)-3-{[2-(3,4-dichlorophenyl)ethyl](2-methylpropyl)amino}-2-hydroxypropoxy]phenyl}methanesulfonamide C22 H30 Cl2 N2 O4 S ZAAZVMJLSXMDHB-IBGZPJMESA-N |  | ||
| NAG Download:Ideal Coordinates CCD File | F [auth A] J [auth B] K [auth B] P [auth C] Q [auth C] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| CL Download:Ideal Coordinates CCD File | H [auth A] I [auth A] M [auth B] N [auth B] O [auth B] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| NA Download:Ideal Coordinates CCD File | G [auth A], R [auth C] | SODIUM ION Na FKNQFGJONOIPTF-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 268.42 | α = 90 |
| b = 59.536 | β = 116.69 |
| c = 146.114 | γ = 90 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data scaling |
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |
| DENZO | data reduction |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of Neurological Disorders and Stroke (NIH/NINDS) | United States | F32NS093753 |
| National Institutes of Health/National Institute of Mental Health (NIH/NIMH) | United States | R01MH085926 |