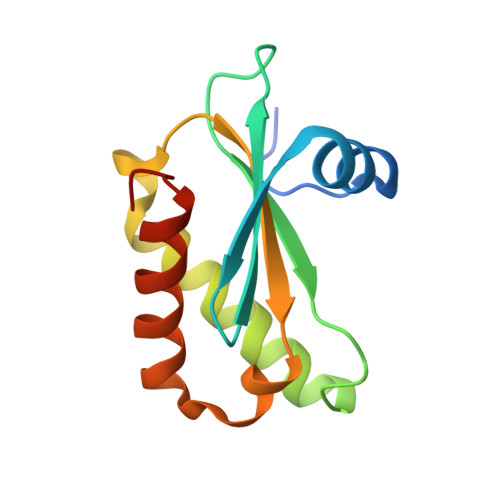
Crystal structure of the ribonuclease-P-protein subunit from Staphylococcus aureus.
Ha, L., Colquhoun, J., Noinaj, N., Das, C., Dunman, P.M., Flaherty, D.P.(2018) Acta Crystallogr F Struct Biol Commun 74: 632-637
- PubMed: 30279314 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1107/S2053230X18011512
- Primary Citation Related Structures:
6D1R - PubMed Abstract:
Staphylococcus aureus ribonuclease-P-protein subunit (RnpA) is a promising antimicrobial target that is a key protein component for two essential cellular processes, RNA degradation and transfer-RNA (tRNA) maturation. The first crystal structure of RnpA from the pathogenic bacterial species, S. aureus, is reported at 2.0 Å resolution. The structure presented maintains key similarities with previously reported RnpA structures from bacteria and archaea, including the highly conserved RNR-box region and aromatic residues in the precursor-tRNA 5'-leader-binding domain. This structure will be instrumental in the pursuit of structure-based designed inhibitors targeting RnpA-mediated RNA processing as a novel therapeutic approach for treating S. aureus infections.
- Department of Medicinal Chemistry and Molecular Pharmacology, College of Pharmacy, Purdue University, West Lafayette, IN 47907, USA.
Organizational Affiliation: