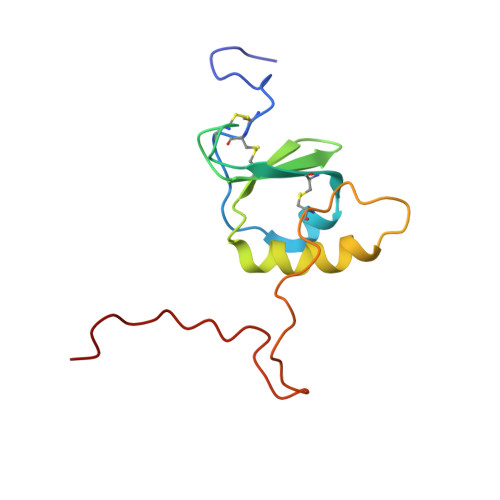
The Solution Structure of CCL28 Reveals Structural Lability that Does Not Constrain Antifungal Activity.
Thomas, M.A., He, J., Peterson, F.C., Huppler, A.R., Volkman, B.F.(2018) J Mol Biology 430: 3266-3282
- PubMed: 29913161 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jmb.2018.06.001
- Primary Citation Related Structures:
6CWS - PubMed Abstract:
The chemokine CCL28 is constitutively expressed in mucosal tissues and is abundant in low-salt mucosal secretions. Beyond its traditional role as a chemoattractant, CCL28 has been shown to act as a potent and broad-spectrum antimicrobial agent with particular efficacy against the commensal fungus and opportunistic pathogen Candida albicans. However, the structural features that allow CCL28 to perform its chemotactic and antimicrobial functions remain unknown. Here, we report the structure of CCL28, solved using nuclear magnetic resonance spectroscopy. CCL28 adopts the canonical chemokine tertiary fold, but also has a disordered C-terminal domain that is partially tethered to the core by a non-conserved disulfide bond. Structure-function analysis reveals that removal of the C-terminal tail reduces the antifungal activity of CCL28 without disrupting its structural integrity. Conversely, removal of the non-conserved disulfide bond destabilizes the tertiary fold of CCL28 without altering its antifungal effects. Moreover, we report that CCL28 unfolds in response to low pH but is stabilized by the presence of salt. To explore the physiologic relevance of the observed structural lability of CCL28, we investigated the effects of pH and salt on the antifungal activity of CCL28 in vitro. We found that low pH enhances the antifungal potency of CCL28, but also that this pH effect is independent of CCL28's tertiary fold. Given its dual role as a chemoattractant and antimicrobial agent, our results suggest that changes in the salt concentration or pH at mucosal sites may fine-tune CCL28's functional repertoire by adjusting the thermostability of its structure.
- Department of Biochemistry, Medical College of Wisconsin, Milwaukee, WI 53226, USA.
Organizational Affiliation: