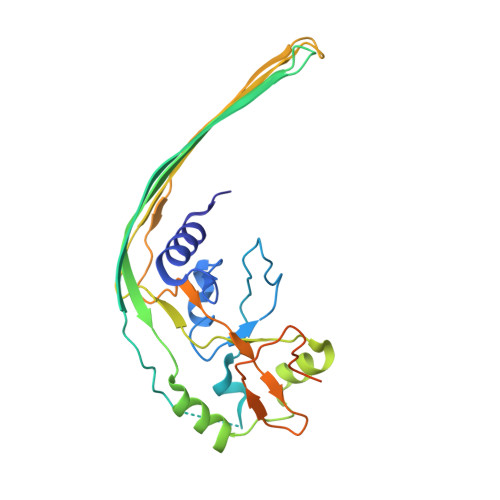
Cryo-EM structure of the gasdermin A3 membrane pore.
Ruan, J., Xia, S., Liu, X., Lieberman, J., Wu, H.(2018) Nature 557: 62-67
- PubMed: 29695864 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41586-018-0058-6
- Primary Citation Related Structures:
6CB8 - PubMed Abstract:
Gasdermins mediate inflammatory cell death after cleavage by caspases or other, unknown enzymes. The cleaved N-terminal fragments bind to acidic membrane lipids to form pores, but the mechanism of pore formation remains unresolved. Here we present the cryo-electron microscopy structures of the 27-fold and 28-fold single-ring pores formed by the N-terminal fragment of mouse GSDMA3 (GSDMA3-NT) at 3.8 and 4.2 Å resolutions, and of a double-ring pore at 4.6 Å resolution. In the 27-fold pore, a 108-stranded anti-parallel β-barrel is formed by two β-hairpins from each subunit capped by a globular domain. We identify a positively charged helix that interacts with the acidic lipid cardiolipin. GSDMA3-NT undergoes radical conformational changes upon membrane insertion to form long, membrane-spanning β-strands. We also observe an unexpected additional symmetric ring of GSDMA3-NT subunits that does not insert into the membrane in the double-ring pore, which may represent a pre-pore state of GSDMA3-NT. These structures provide a basis that explains the activities of several mutant gasdermins, including defective mutants that are associated with cancer.
- Program in Cellular and Molecular Medicine, Boston Children's Hospital, Boston, MA, USA.
Organizational Affiliation: