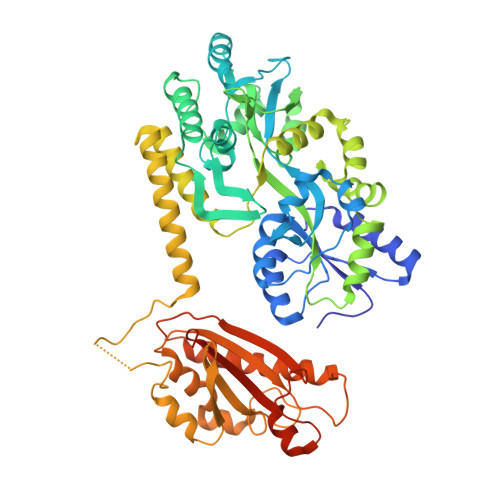
Crystal structure of the periplasmic domain of TssL, a key membrane component of Type VI secretion system.
Wang, X., Sun, B., Xu, M., Qiu, S., Xu, D., Ran, T., He, J., Wang, W.(2018) Int J Biol Macromol 120: 1474-1479
- PubMed: 30266644 Search on PubMed
- DOI: https://doi.org/10.1016/j.ijbiomac.2018.09.166
- Primary Citation Related Structures:
6AEO - PubMed Abstract:
Type VI secretion system (T6SS), as a macromolecular system, is commonly found in Gram-negative bacteria and responsible for exporting effectors. T6SS consists of 13 core proteins. TssL is a component of the membrane complex and plays a pivotal role in T6SS. Here, we report the crystal structure of the C-terminal periplasmic domain of TssL (TssL Cter ) from Serratia marcescens FS14. The TssL Cter (310-503) contain a five-stranded anti-parallel β-sheet flanked by five α-helices and a short N-terminal helix. Structural comparisons revealed that it belongs to the OmpA-like family with a remarked difference in the conformation of the loop3-5. In OmpA-like family, the corresponding loop is located close to loop2-3, forming a cavity with a small opening together with the longest α5, whereas in TssL Cter , loop3-5 flipped away from this cavity region. In addition, significant differences in the peptidoglycan (PG) binding site suggest that big conformational change must take place to accomplish the PG binding for TssL Cter . Furthermore, a long flexible loop between helices α1 and α2 is unique in TssL. TssL would have a big conformational change during the delivery of the Hcp needle and effectors. So we speculate that the long flexible endows TssL the adaptation of its evolutionary new function.
- Key Laboratory of Microbiological Engineering of Agricultural Environment of Ministry of Agriculture, Department of Microbiology, College of Life Sciences, Nanjing Agricultural University, Nanjing, China.
Organizational Affiliation: