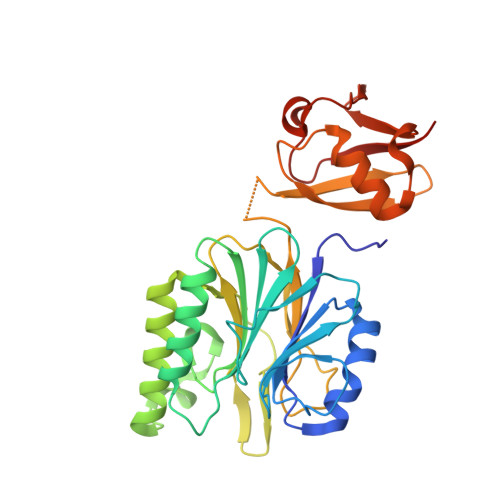
Crystal Structure Determination of Ubiquitin by Fusion to a Protein That Forms a Highly Porous Crystal Lattice
Maita, N.(2018) J Am Chem Soc 140: 13546-13549
- PubMed: 30299944 Search on PubMed
- DOI: https://doi.org/10.1021/jacs.8b07512
- Primary Citation Related Structures:
6A42, 6A43, 6A44 - PubMed Abstract:
The protein crystallization process requires screening of a large number of conditions using a large quantity of high-purity protein, which makes crystal structure analysis difficult. Thus, the development of easy and versatile protein crystallization techniques is both extremely desirable and highly challenging. Here I demonstrate the crystallization and structure determination of ubiquitin by genetic fusion to the highly porous honeycomb lattice of R1EN. I successfully crystallized and collected X-ray data from three R1EN-ubiquitin constructs with various linker lengths under the same conditions as the original R1EN. The crystals diffracted to 1.7-2.4 Å resolution, and the ubiquitin structures were determined with results almost identical to the previously published structure. Moreover, the ubiquitin structure could be solved by molecular replacement using R1EN alone. This method may reduce the effort required for crystallization screening and is applicable to de novo protein structure determination.
- Division of Disease Proteomics, Institute of Advanced Medical Sciences , Tokushima University , 3-18-15 Kuramoto-cho , Tokushima 770-8503 , Japan.
Organizational Affiliation: