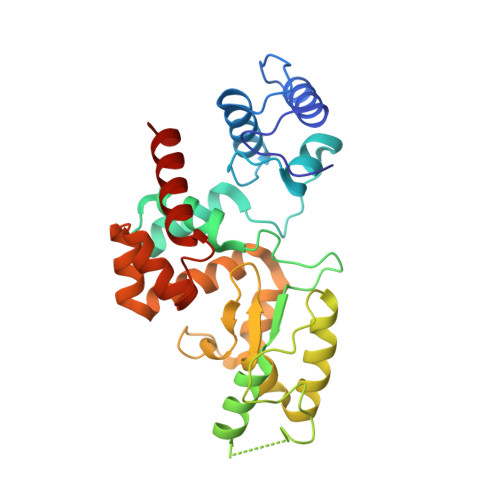
Modular endolysin of Burkholderia AP3 phage has the largest lysozyme-like catalytic subunit discovered to date and no catalytic aspartate residue.
Maciejewska, B., Zrubek, K., Espaillat, A., Wisniewska, M., Rembacz, K.P., Cava, F., Dubin, G., Drulis-Kawa, Z.(2017) Sci Rep 7: 14501-14501
- PubMed: 29109551 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41598-017-14797-9
- Primary Citation Related Structures:
5NM7 - PubMed Abstract:
Endolysins are peptidoglycan-degrading enzymes utilized by bacteriophages to release the progeny from bacterial cells. The lytic properties of phage endolysins make them potential antibacterial agents for medical and industrial applications. Here, we present a comprehensive characterization of phage AP3 modular endolysin (AP3gp15) containing cell wall binding domain and an enzymatic domain (DUF3380 by BLASTP), both widespread and conservative. Our structural analysis demonstrates the low similarity of an enzymatic domain to known lysozymes and an unusual catalytic centre characterized by only a single glutamic acid residue and no aspartic acid. Thus, our findings suggest distinguishing a novel class of muralytic enzymes having the activity and catalytic centre organization of DUF3380. The lack of amino acid sequence homology between AP3gp15 and other known muralytic enzymes may reflect the evolutionary convergence of analogous glycosidases. Moreover, the broad antibacterial spectrum, lack of cytotoxic effect on human cells and the stability characteristics of AP3 endolysin advocate for its future application development.
- Institute of Genetics and Microbiology, University of Wroclaw, Przybyszewskiego 63/77, 51-148, Wroclaw, Poland.
Organizational Affiliation: