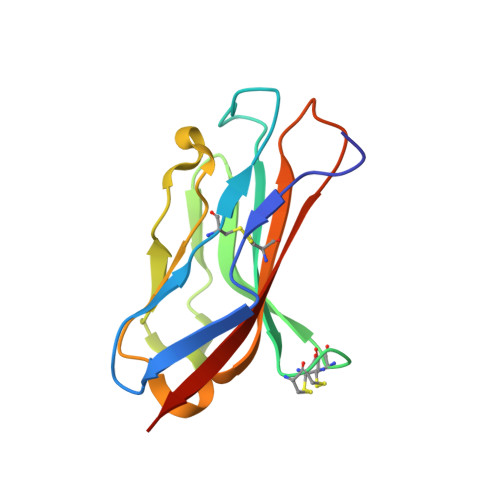
Binary architecture of the Nav1.2-beta 2 signaling complex.
Das, S., Gilchrist, J., Bosmans, F., Van Petegem, F.(2016) Elife 5
- PubMed: 26894959 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.7554/eLife.10960
- Primary Citation Related Structures:
5FDY, 5FEB - PubMed Abstract:
To investigate the mechanisms by which β-subunits influence Nav channel function, we solved the crystal structure of the β2 extracellular domain at 1.35Å. We combined these data with known bacterial Nav channel structural insights and novel functional studies to determine the interactions of specific residues in β2 with Nav1.2. We identified a flexible loop formed by (72)Cys and (75)Cys, a unique feature among the four β-subunit isoforms. Moreover, we found that (55)Cys helps to determine the influence of β2 on Nav1.2 toxin susceptibility. Further mutagenesis combined with the use of spider toxins reveals that (55)Cys forms a disulfide bond with (910)Cys in the Nav1.2 domain II pore loop, thereby suggesting a 1:1 stoichiometry. Our results also provide clues as to which disulfide bonds are formed between adjacent Nav1.2 (912/918)Cys residues. The concepts emerging from this work will help to form a model reflecting the β-subunit location in a Nav channel complex.
- Department of Biochemistry and Molecular Biology, Life Sciences Institute, University of British Columbia, Vancouver, Canada.
Organizational Affiliation: