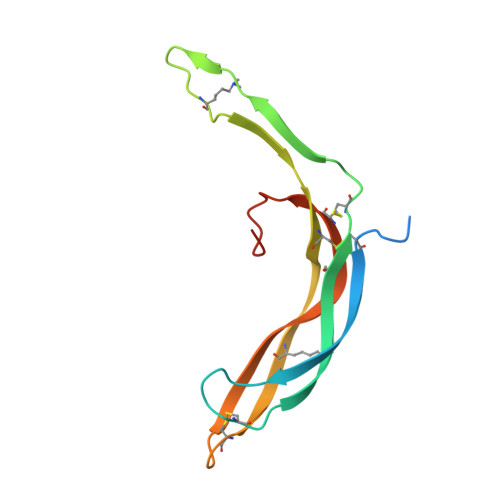
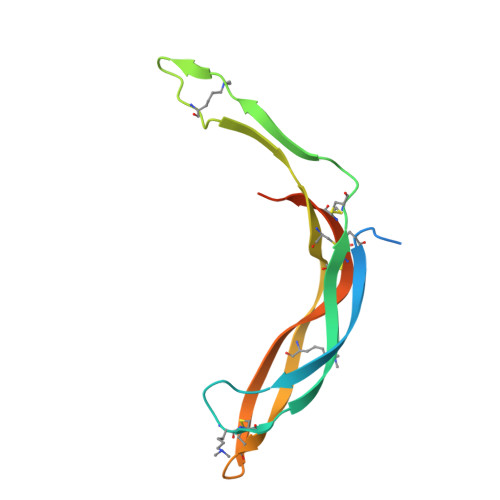
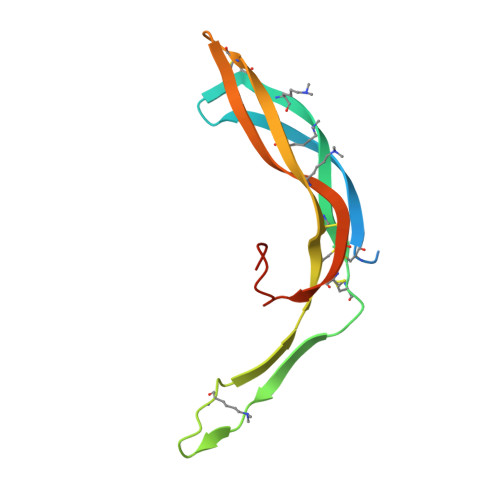
Structure and functional properties of Norrin mimic Wnt for signalling with Frizzled4, Lrp5/6, and proteoglycan.
Chang, T.H., Hsieh, F.L., Zebisch, M., Harlos, K., Elegheert, J., Jones, E.Y.(2015) Elife 4: e06554
- PubMed: 26158506 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.7554/eLife.06554
- Primary Citation Related Structures:
5BPB, 5BPQ, 5BPU, 5BQ8, 5BQB, 5BQC, 5BQE - PubMed Abstract:
Wnt signalling regulates multiple processes including angiogenesis, inflammation, and tumorigenesis. Norrin (Norrie Disease Protein) is a cystine-knot like growth factor. Although unrelated to Wnt, Norrin activates the Wnt/β-catenin pathway. Signal complex formation involves Frizzled4 (Fz4), low-density lipoprotein receptor related protein 5/6 (Lrp5/6), Tetraspanin-12 and glycosaminoglycans (GAGs). Here, we report crystallographic and small-angle X-ray scattering analyses of Norrin in complex with Fz4 cysteine-rich domain (Fz4CRD), of this complex bound with GAG analogues, and of unliganded Norrin and Fz4CRD. Our structural, biophysical and cellular data, map Fz4 and putative Lrp5/6 binding sites to distinct patches on Norrin, and reveal a GAG binding site spanning Norrin and Fz4CRD. These results explain numerous disease-associated mutations. Comparison with the Xenopus Wnt8-mouse Fz8CRD complex reveals Norrin mimics Wnt for Frizzled recognition. The production and characterization of wild-type and mutant Norrins reported here open new avenues for the development of therapeutics to combat abnormal Norrin/Wnt signalling.
- Division of Structural Biology, Wellcome Trust Centre for Human Genetics, University of Oxford, Oxford, United Kingdom.
Organizational Affiliation: