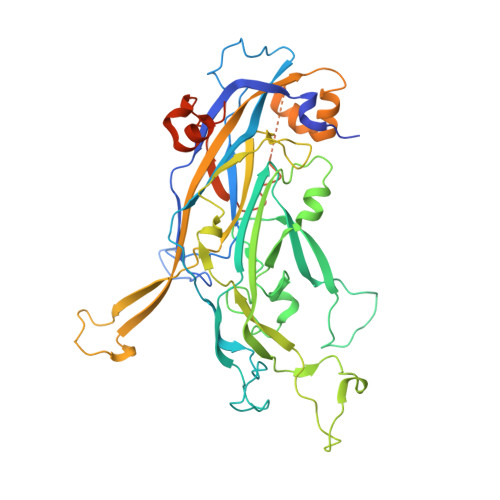
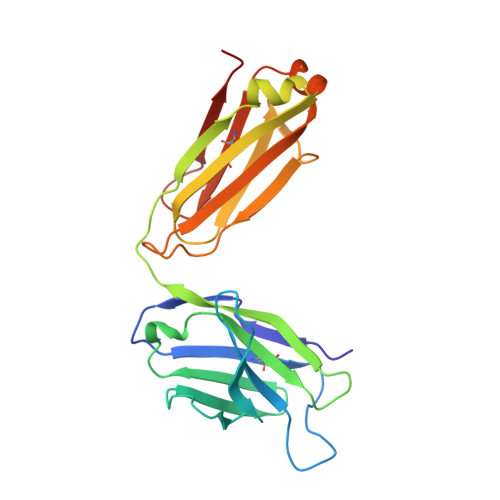
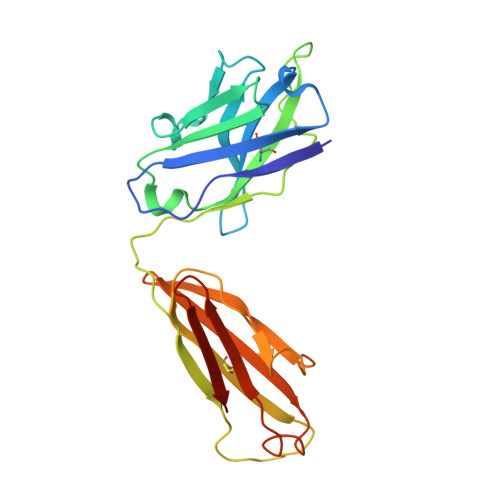
Crystal Structures of Two Immune Complexes Identify Determinants for Viral Infectivity and Type-Specific Neutralization of Human Papillomavirus.
Li, Z., Wang, D., Gu, Y., Song, S., He, M., Shi, J., Liu, X., Wei, S., Li, J., Yu, H., Zheng, Q., Yan, X., Baker, T.S., Zhang, J., McLellan, J.S., Li, S., Xia, N.(2017) mBio 8
- PubMed: 28951471 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1128/mBio.00787-17
- Primary Citation Related Structures:
5Y9C, 5Y9E, 5Y9F - PubMed Abstract:
Persistent, high-risk human papillomavirus (HPV) infection is the primary cause of cervical cancer. Neutralizing antibodies elicited by L1-only virus-like particles (VLPs) can block HPV infection; however, the lack of high-resolution structures has limited our understanding of the mode of virus infection and the requirement for type specificity at the molecular level. Here, we describe two antibodies, A12A3 and 28F10, that specifically bind to and neutralize HPV58 and HPV59, respectively, through two distinct binding stoichiometries. We show that the epitopes of A12A3 are clustered in the DE loops of two adjacent HPV58 L1 monomers, whereas 28F10 recognizes the HPV59 FG loop of a single monomer. Via structure-based mutagenesis and analysis of antibody binding, we further identified the residues HPV58 D154, S168, and N170 and HPV59 M267, Q270, E273, Y276, K278, and R283, which play critical roles in virus infection. By substituting these strategic epitope residues into other HPV genotypes, we could then redirect the type-specific binding of the antibodies to these genotypes, thus highlighting the importance of these specific residues, HPV58 R161, S168, and N308 and HPV59 Q270, E273, and D281. Overall, our findings provide molecular insights into potential structural determinants of HPV required for infectivity and type specificity. IMPORTANCE High-risk human papillomaviruses (HPVs) are considered the major causative pathogens of cancers that affect epithelial mucosa, such as cervical cancer. However, because of the lack of high-resolution structural information on the sites of neutralization, we have yet to determine the precise mode of HPV infection and how different types of HPV cause infection. Our crystal structures in this study have uncovered discrete binding stoichiometries for two different antibodies. We show that one A12A3 Fab binds to the center of one HPV58 pentamer, whereas five 28F10 Fabs bind along the top fringe of one HPV59 pentamer. Furthermore, through targeted epitope analysis, we show that 6 to 7 discontinuous residues of the L1 major capsid protein of HPV are determinants, at least in part, for virus infection and type specificity. This knowledge will help us to unravel the process of HPV infection and can potentially be used to drive the development of therapeutics that target neutralization-sensitive sites.
- State Key Laboratory of Molecular Vaccinology and Molecular Diagnostics, School of Life Sciences, Xiamen University, Xiamen, China.
Organizational Affiliation: