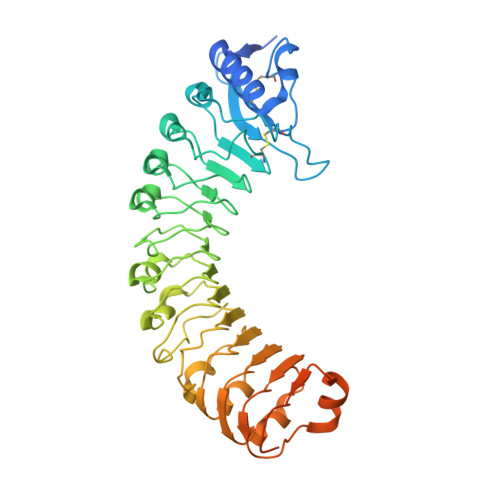
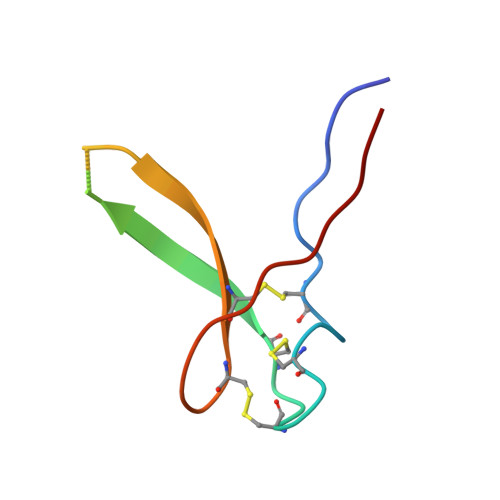
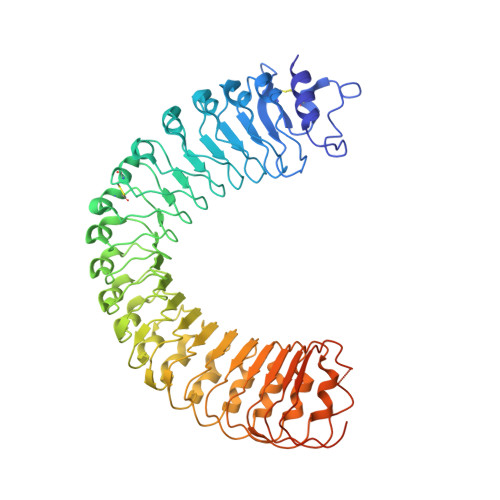
A receptor-like protein acts as a specificity switch for the regulation of stomatal development.
Lin, G., Zhang, L., Han, Z., Yang, X., Liu, W., Li, E., Chang, J., Qi, Y., Shpak, E.D., Chai, J.(2017) Genes Dev 31: 927-938
- PubMed: 28536146 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1101/gad.297580.117
- Primary Citation Related Structures:
5XJO, 5XJX, 5XKJ, 5XKN - PubMed Abstract:
Stomata are microscopic openings that allow for the exchange of gases between plants and the environment. In Arabidopsis , stomatal patterning is specified by the ERECTA family (ERf) receptor kinases (RKs), the receptor-like protein (RLP) TOO MANY MOUTHS (TMM), and EPIDERMAL PATTERNING FACTOR (EPF) peptides. Here we show that TMM and ER or ER-LIKE1 (ERL1) form constitutive complexes, which recognize EPF1 and EPF2, but the single ERfs do not. TMM interaction with ERL1 creates a binding pocket for recognition of EPF1 and EPF2, indicating that the constitutive TMM-ERf complexes function as the receptors of EPF1 and EPF2. EPFL9 competes with EPF1 and EPF2 for binding to the ERf-TMM complex. EPFL4 and EPFL6, however, are recognized by the single ERfs without the requirement of TMM. In contrast to EPF1,2, the interaction of EPFL4,6 with an ERf is greatly reduced in the presence of TMM. Taken together, our data demonstrate that TMM dictates the specificity of ERfs for the perception of different EPFs, thus functioning as a specificity switch for the regulation of the activities of ERfs.
- Innovation Center for Structural Biology, Tsinghua-Peking Joint Center for Life Sciences, School of Life Sciences, Tsinghua University, Beijing 100084, China.
Organizational Affiliation: