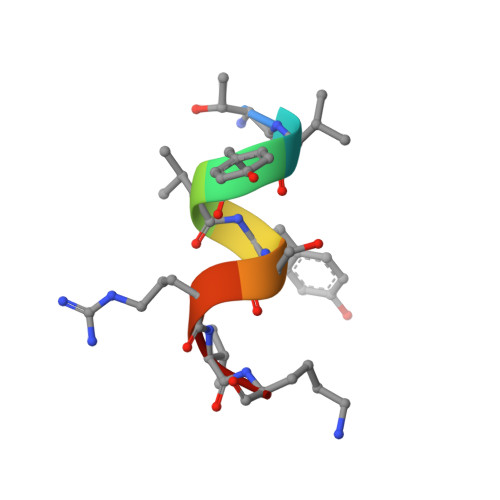
Structural insights of a self-assembling 9-residue peptide from the C-terminal tail of the SARS corona virus E-protein in DPC and SDS micelles: A combined high and low resolution spectroscopic study.
Ghosh, A., Bhattacharyya, D., Bhunia, A.(2018) Biochim Biophys Acta Biomembr 1860: 335-346
- PubMed: 29038024 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.bbamem.2017.10.015
- Primary Citation Related Structures:
5XER, 5XES - PubMed Abstract:
In recent years, several studies based on the interaction of self-assembling short peptides derived from viroporins with model membranes, have improved our understanding of the molecular mechanism of corona virus (CoV) infection under physiological conditions. In this study, we have characterized the mechanism of membrane interaction of a short, 9-residue peptide TK9 (T 55 VYVYSRVK 63 ) that had been derived from the carboxyl terminal of the Severe Acute Respiratory Syndrome (SARS) corona virus (SARS CoV) envelope (E) protein. The peptide has been studied for its physical changes in the presence of both zwitterionic DPC and negatively charged SDS model membrane micelles, respectively, with the help of a battery of biophysical techniques including two-dimensional solution state NMR spectroscopy. Interestingly, in both micellar environments, TK9 adopted an alpha helical conformation; however, the helical propensities were much higher in the case of DPC compared to those of SDS micelle, suggesting that TK9 has more specificity towards eukaryotic cell membrane than the bacterial cell membrane. The orientation of the peptide TK9 also varies in the different micellar environments. The peptide's affinity was further manifested by its pronounced membrane disruption ability towards the mammalian compared to the bacterial membrane mimic. Collectively, the in-depth structural information on the interaction of TK9 with different membrane environments explains the host specificity and membrane orientation owing to subsequent membrane disruption implicated in the viral pathogenesis.
- Department of Biophysics, Bose Institute, P-1/12 CIT Scheme VII (M), Kolkata 700054, India.
Organizational Affiliation: