2.05 A resolution structure of IpaD from Shigella flexneri in complex with single-domain antibody 20ipaD
Barta, M.L., Lovell, S., Battaile, K.P., Picking, W.D., Picking, W.L.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Invasin IpaD | 289 | Shigella flexneri | Mutation(s): 1 Gene Names: ipaD, CP0126 | 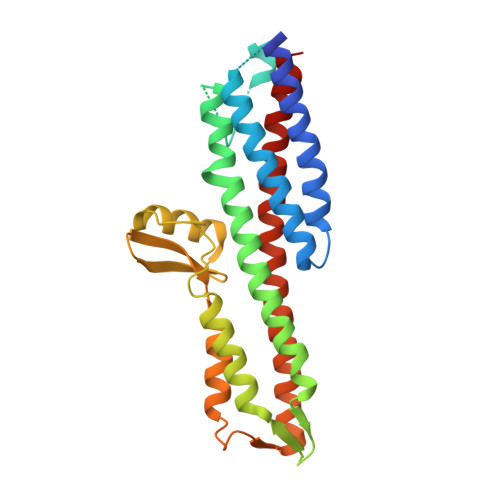 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P18013 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Single-domain antibody 20ipaD | 151 | Vicugna pacos | Mutation(s): 0 | 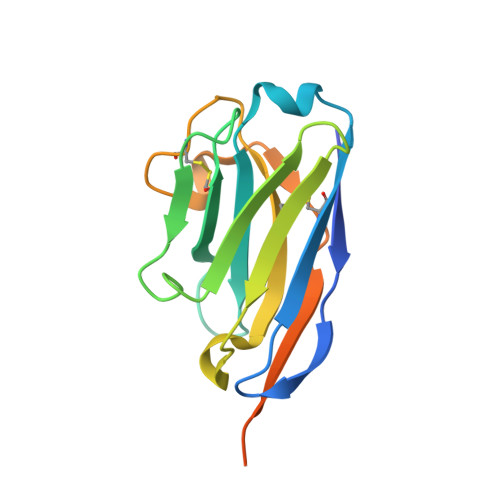 | |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 58.642 | α = 90 |
| b = 83.507 | β = 90 |
| c = 181.609 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| Aimless | data scaling |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |
| PHASER | phasing |