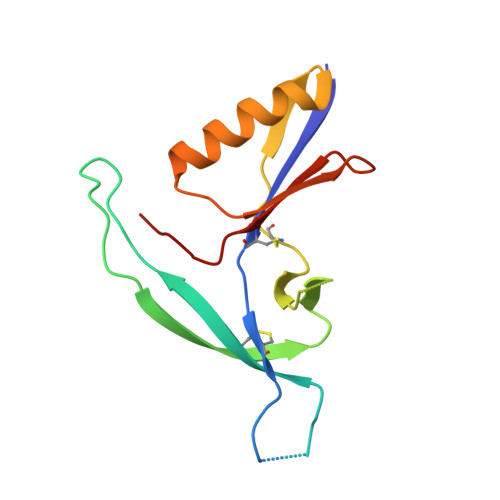
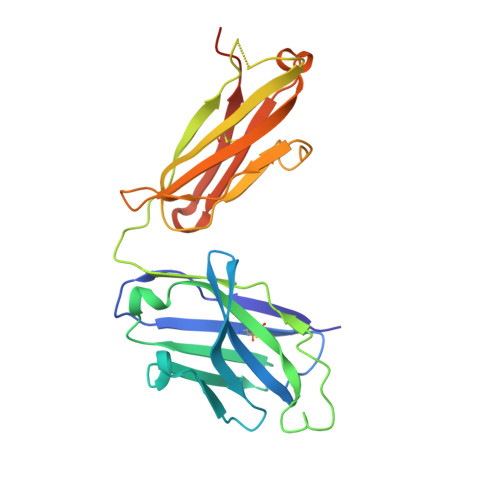
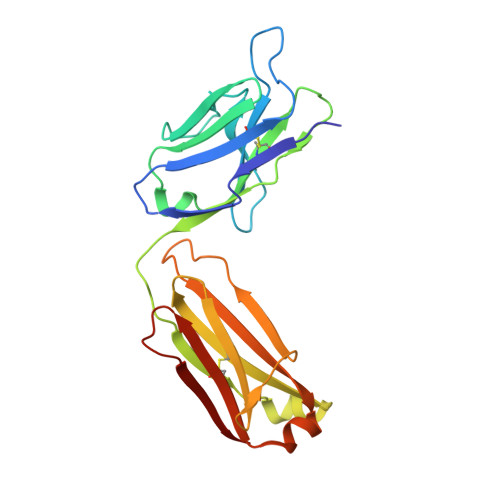
Virus-like Particles Identify an HIV V1V2 Apex-Binding Neutralizing Antibody that Lacks a Protruding Loop.
Cale, E.M., Gorman, J., Radakovich, N.A., Crooks, E.T., Osawa, K., Tong, T., Li, J., Nagarajan, R., Ozorowski, G., Ambrozak, D.R., Asokan, M., Bailer, R.T., Bennici, A.K., Chen, X., Doria-Rose, N.A., Druz, A., Feng, Y., Joyce, M.G., Louder, M.K., O'Dell, S., Oliver, C., Pancera, M., Connors, M., Hope, T.J., Kepler, T.B., Wyatt, R.T., Ward, A.B., Georgiev, I.S., Kwong, P.D., Mascola, J.R., Binley, J.M.(2017) Immunity 46: 777-791.e10
- PubMed: 28514685 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.immuni.2017.04.011
- Primary Citation Related Structures:
5EWI, 5VGJ - PubMed Abstract:
Most HIV-1-specific neutralizing antibodies isolated to date exhibit unusual characteristics that complicate their elicitation. Neutralizing antibodies that target the V1V2 apex of the HIV-1 envelope (Env) trimer feature unusually long protruding loops, which enable them to penetrate the HIV-1 glycan shield. As antibodies with loops of requisite length are created through uncommon recombination events, an alternative mode of apex binding has been sought. Here, we isolated a lineage of Env apex-directed neutralizing antibodies, N90-VRC38.01-11, by using virus-like particles and conformationally stabilized Env trimers as B cell probes. A crystal structure of N90-VRC38.01 with a scaffolded V1V2 revealed a binding mode involving side-chain-to-side-chain interactions that reduced the distance the antibody loop must traverse the glycan shield, thereby facilitating V1V2 binding via a non-protruding loop. The N90-VRC38 lineage thus identifies a solution for V1V2-apex binding that provides a more conventional B cell pathway for vaccine design.
- Vaccine Research Center, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD 20892, USA.
Organizational Affiliation: