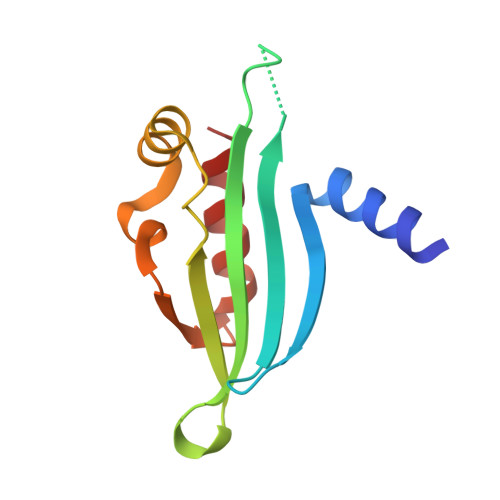
Recruitment of a SUMO isopeptidase to rDNA stabilizes silencing complexes by opposing SUMO targeted ubiquitin ligase activity.
Liang, J., Singh, N., Carlson, C.R., Albuquerque, C.P., Corbett, K.D., Zhou, H.(2017) Genes Dev 31: 802-815
- PubMed: 28487408 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1101/gad.296145.117
- Primary Citation Related Structures:
5V1A, 5V3N - PubMed Abstract:
Post-translational modification by SUMO (small ubiquitin-like modifier) plays important but still poorly understood regulatory roles in eukaryotic cells, including as a signal for ubiquitination by SUMO targeted ubiquitin ligases (STUbLs). Here, we delineate the molecular mechanisms for SUMO-dependent control of ribosomal DNA (rDNA) silencing through the opposing actions of a STUbL (Slx5:Slx8) and a SUMO isopeptidase (Ulp2). We identify a conserved region in the Ulp2 C terminus that mediates its specificity for rDNA-associated proteins and show that this region binds directly to the rDNA-associated protein Csm1. Two crystal structures show that Csm1 interacts with Ulp2 and one of its substrates, the rDNA silencing protein Tof2, through adjacent conserved interfaces in its C-terminal domain. Disrupting Csm1's interaction with either Ulp2 or Tof2 dramatically reduces rDNA silencing and causes a marked drop in Tof2 abundance, suggesting that Ulp2 promotes rDNA silencing by opposing STUbL-mediated degradation of silencing proteins. Tof2 abundance is rescued by deletion of the STUbL SLX5 or disruption of its SUMO-interacting motifs, confirming that Tof2 is targeted for degradation in a SUMO- and STUbL-dependent manner. Overall, our results demonstrate how the opposing actions of a localized SUMO isopeptidase and a STUbL regulate rDNA silencing by controlling the abundance of a key rDNA silencing protein, Tof2.
- Ludwig Institute for Cancer Research, San Diego Branch, La Jolla, California 92093, USA.
Organizational Affiliation: