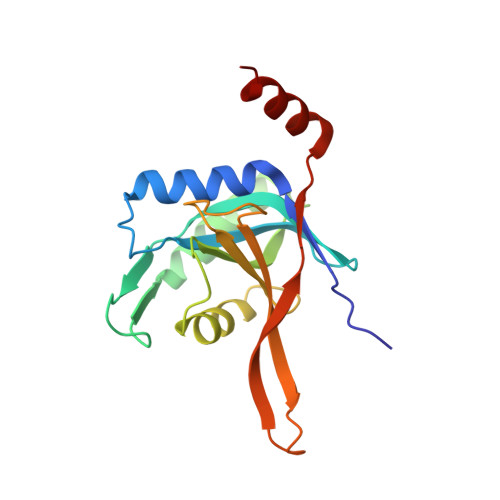
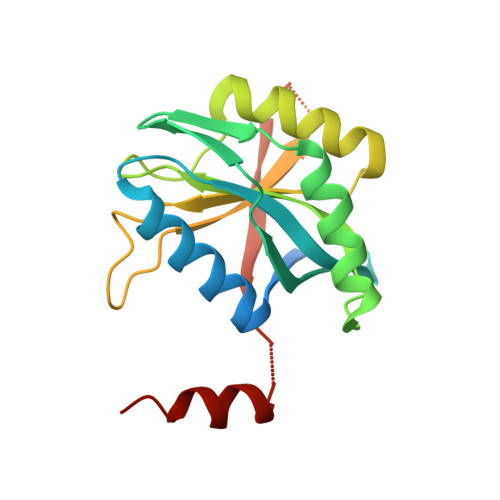
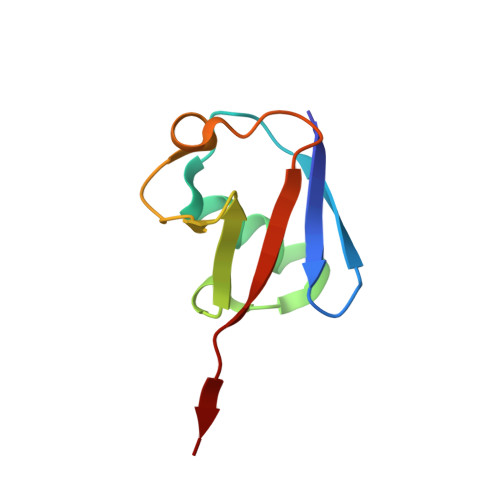
An AAA Motor-Driven Mechanical Switch in Rpn11 Controls Deubiquitination at the 26S Proteasome.
Worden, E.J., Dong, K.C., Martin, A.(2017) Mol Cell 67: 799-811.e8
- PubMed: 28844860 Search on PubMed
- DOI: https://doi.org/10.1016/j.molcel.2017.07.023
- Primary Citation Related Structures:
5U4P, 5W83 - PubMed Abstract:
Poly-ubiquitin chains direct protein substrates to the 26S proteasome, where they are removed by the deubiquitinase Rpn11 during ATP-dependent substrate degradation. Rapid deubiquitination is required for efficient degradation but must be restricted to committed substrates that are engaged with the ATPase motor to prevent premature ubiquitin chain removal and substrate escape. Here we reveal the ubiquitin-bound structure of Rpn11 from S. cerevisiae and the mechanisms for mechanochemical coupling of substrate degradation and deubiquitination. Ubiquitin binding induces a conformational switch of Rpn11's Insert-1 loop from an inactive closed state to an active β hairpin. This switch is rate-limiting for deubiquitination and strongly accelerated by mechanical substrate translocation into the AAA+ motor. Deubiquitination by Rpn11 and ubiquitin unfolding by the ATPases are in direct competition. The AAA+ motor-driven acceleration of Rpn11 is therefore important to ensure that poly-ubiquitin chains are removed only from committed substrates and fast enough to prevent their co-degradation.
- Department of Molecular and Cell Biology, University of California, Berkeley, Berkeley, CA 94720, USA; California Institute for Quantitative Biosciences, University of California, Berkeley, Berkeley, CA 94720, USA.
Organizational Affiliation: