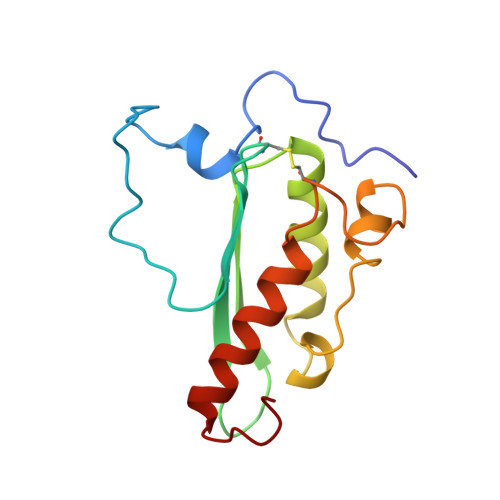
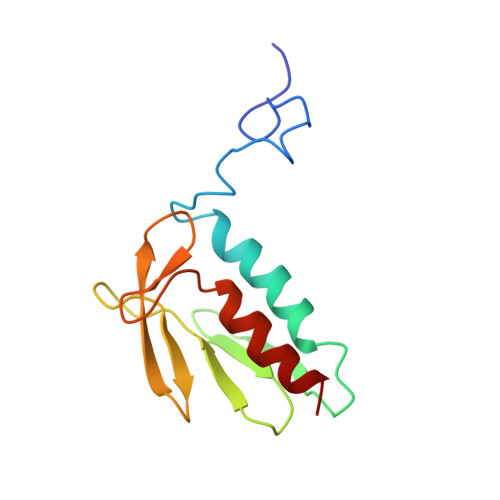
Zinc and the iron donor frataxin regulate oligomerization of the scaffold protein to form new Fe-S cluster assembly centers.
Galeano, B.K., Ranatunga, W., Gakh, O., Smith, D.Y., Thompson, J.R., Isaya, G.(2017) Metallomics 9: 773-801
- PubMed: 28548666
- DOI: https://doi.org/10.1039/c7mt00089h
- Primary Citation Related Structures:
5TRE - PubMed Abstract:
Early studies of the bacterial Fe-S cluster assembly system provided structural details for how the scaffold protein and the cysteine desulfurase interact. This work and additional work on the yeast and human systems elucidated a conserved mechanism for sulfur donation but did not provide any conclusive insights into the mechanism for iron delivery from the iron donor, frataxin, to the scaffold. We previously showed that oligomerization is a mechanism by which yeast frataxin (Yfh1) can promote assembly of the core machinery for Fe-S cluster synthesis both in vitro and in cells, in such a manner that the scaffold protein, Isu1, can bind to Yfh1 independent of the presence of the cysteine desulfurase, Nfs1. Here, in the absence of Yfh1, Isu1 was found to exist in two forms, one mostly monomeric with limited tendency to dimerize, and one with a strong propensity to oligomerize. Whereas the monomeric form is stabilized by zinc, the loss of zinc promotes formation of dimer and higher order oligomers. However, upon binding to oligomeric Yfh1, both forms take on a similar symmetrical trimeric configuration that places the Fe-S cluster coordinating residues of Isu1 in close proximity of iron-binding residues of Yfh1. This configuration is suitable for docking of Nfs1 in a manner that provides a structural context for coordinate iron and sulfur donation to the scaffold. Moreover, distinct structural features suggest that in physiological conditions the zinc-regulated abundance of monomeric vs. oligomeric Isu1 yields [Yfh1]·[Isu1] complexes with different Isu1 configurations that afford unique functional properties for Fe-S cluster assembly and delivery.
- Department of Pediatric & Adolescent Medicine, Mayo Clinic, Rochester, Minnesota, USA. gakh.oleksandr@mayo.edu isaya@mayo.edu.
Organizational Affiliation: