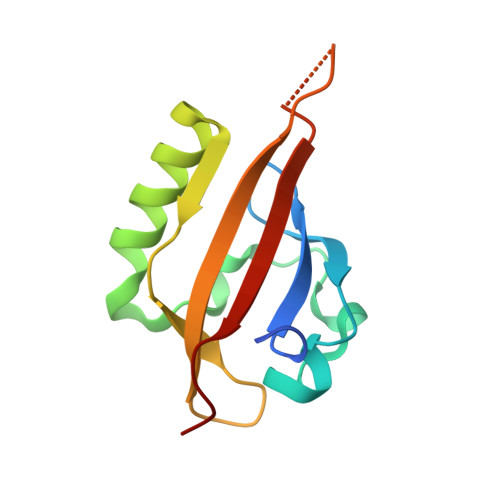
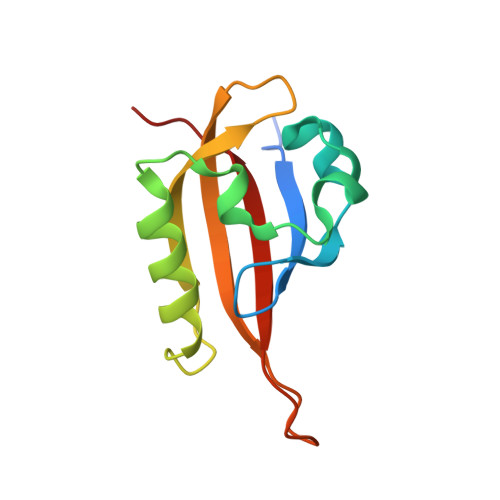
A Small-Molecule Antagonist of HIF2 alpha Is Efficacious in Preclinical Models of Renal Cell Carcinoma.
Wallace, E.M., Rizzi, J.P., Han, G., Wehn, P.M., Cao, Z., Du, X., Cheng, T., Czerwinski, R.M., Dixon, D.D., Goggin, B.S., Grina, J.A., Halfmann, M.M., Maddie, M.A., Olive, S.R., Schlachter, S.T., Tan, H., Wang, B., Wang, K., Xie, S., Xu, R., Yang, H., Josey, J.A.(2016) Cancer Res 76: 5491-5500
- PubMed: 27635045 Search on PubMed
- DOI: https://doi.org/10.1158/0008-5472.CAN-16-0473
- Primary Citation Related Structures:
5TBM - PubMed Abstract:
More than 90% of clear cell renal cell carcinomas (ccRCC) exhibit inactivation of the von Hippel-Lindau (pVHL) tumor suppressor, establishing it as the major underlying cause of this malignancy. pVHL inactivation results in stabilization of the hypoxia-inducible transcription factors, HIF1α and HIF2α, leading to expression of a genetic program essential for the initiation and progression of ccRCC. Herein, we describe the potent, selective, and orally active small-molecule inhibitor PT2385 as a specific antagonist of HIF2α that allosterically blocks its dimerization with the HIF1α/2α transcriptional dimerization partner ARNT/HIF1β. PT2385 inhibited the expression of HIF2α-dependent genes, including VEGF-A, PAI-1, and cyclin D1 in ccRCC cell lines and tumor xenografts. Treatment of tumor-bearing mice with PT2385 caused dramatic tumor regressions, validating HIF2α as a pivotal oncogenic driver in ccRCC. Notably, unlike other anticancer agents that inhibit VEGF receptor signaling, PT2385 exhibited no adverse effect on cardiovascular performance. Thus, PT2385 represents a novel class of therapeutics for the treatment of RCC with potent preclincal efficacy as well as improved tolerability relative to current agents that target the VEGF pathway. Cancer Res; 76(18); 5491-500. ©2016 AACR.
- Peloton Therapeutics, Inc., Dallas, Texas. eli.wallace@pelotontx.com.
Organizational Affiliation: