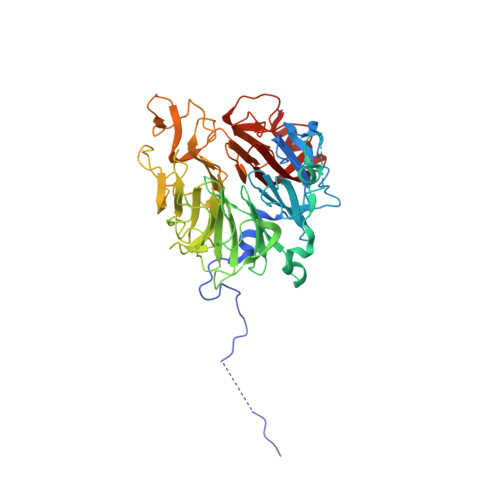
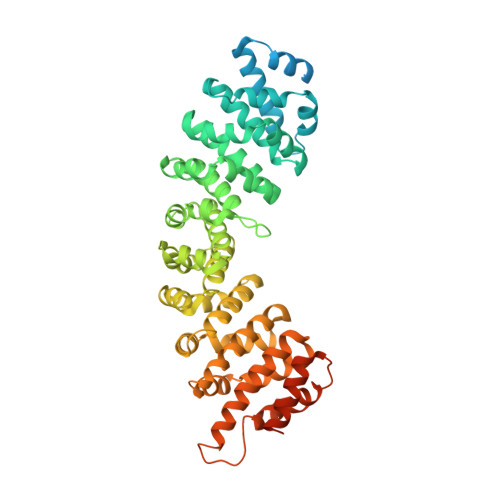
Three-dimensional context rather than NLS amino acid sequence determines importin alpha subtype specificity for RCC1.
Sankhala, R.S., Lokareddy, R.K., Begum, S., Pumroy, R.A., Gillilan, R.E., Cingolani, G.(2017) Nat Commun 8: 979-979
- PubMed: 29042532
- DOI: https://doi.org/10.1038/s41467-017-01057-7
- Primary Citation Related Structures:
5T94, 5TBK - PubMed Abstract:
Active nuclear import of Ran exchange factor RCC1 is mediated by importin α3. This pathway is essential to generate a gradient of RanGTP on chromatin that directs nucleocytoplasmic transport, mitotic spindle assembly and nuclear envelope formation. Here we identify the mechanisms of importin α3 selectivity for RCC1. We find this isoform binds RCC1 with one order of magnitude higher affinity than the generic importin α1, although the two isoforms share an identical NLS-binding groove. Importin α3 uses its greater conformational flexibility to wedge the RCC1 β-propeller flanking the NLS against its lateral surface, preventing steric clashes with its Armadillo-core. Removing the β-propeller, or inserting a linker between NLS and β-propeller, disrupts specificity for importin α3, demonstrating the structural context rather than NLS sequence determines selectivity for isoform 3. We propose importin α3 evolved to recognize topologically complex NLSs that lie next to bulky domains or are masked by quaternary structures.Importin α3 facilitates the nuclear transport of the Ran guanine nucleotide exchange factor RCC1. Here the authors reveal the molecular basis for the selectivity of RCC1 for importin α3 vs the generic importin α1 and discuss the evolution of importin α isoforms.
- Department of Biochemistry and Molecular Biology, Thomas Jefferson University, 233 South 10th Street, Philadelphia, PA, 19107, USA.
Organizational Affiliation: