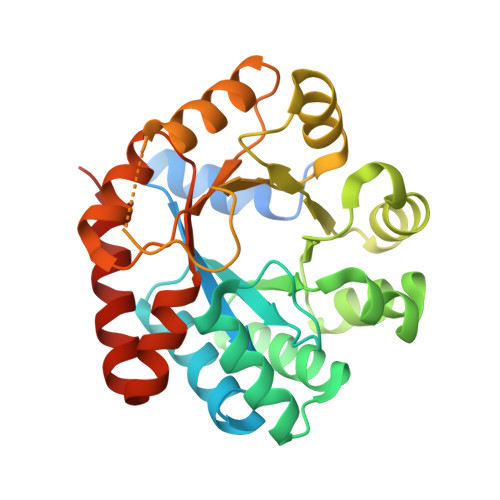
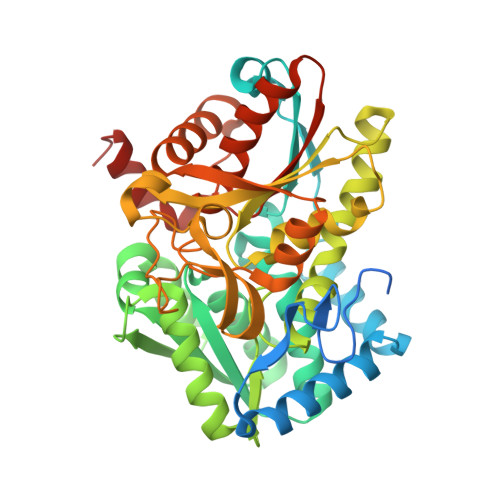
Inhibiting mycobacterial tryptophan synthase by targeting the inter-subunit interface.
Abrahams, K.A., Cox, J.A.G., Futterer, K., Rullas, J., Ortega-Muro, F., Loman, N.J., Moynihan, P.J., Perez-Herran, E., Jimenez, E., Esquivias, J., Barros, D., Ballell, L., Alemparte, C., Besra, G.S.(2017) Sci Rep 7: 9430-9430
- PubMed: 28842600 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41598-017-09642-y
- Primary Citation Related Structures:
5OCW - PubMed Abstract:
Drug discovery efforts against the pathogen Mycobacterium tuberculosis (Mtb) have been advanced through phenotypic screens of extensive compound libraries. Such a screen revealed sulfolane 1 and indoline-5-sulfonamides 2 and 3 as potent inhibitors of mycobacterial growth. Optimization in the sulfolane series led to compound 4, which has proven activity in an in vivo murine model of Mtb infection. Here we identify the target and mode of inhibition of these compounds based on whole genome sequencing of spontaneous resistant mutants, which identified mutations locating to the essential α- and β-subunits of tryptophan synthase. Over-expression studies confirmed tryptophan synthase as the biological target. Biochemical techniques probed the mechanism of inhibition, revealing the mutant enzyme complex incurs a fitness cost but does not prevent inhibitor binding. Mapping of the resistance conferring mutations onto a low-resolution crystal structure of Mtb tryptophan synthase showed they locate to the interface between the α- and β-subunits. The discovery of anti-tubercular agents inhibiting tryptophan synthase highlights the therapeutic potential of this enzyme and draws attention to the prospect of other amino acid biosynthetic pathways as future Mtb drug targets.
- Institute of Microbiology and Infection, School of Biosciences, University of Birmingham, Edgbaston, Birmingham, B15 2TT, UK.
Organizational Affiliation: