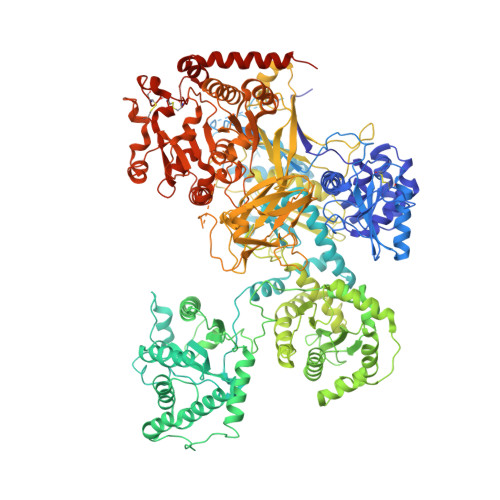
Interdomain conformational flexibility underpins the activity of UGGT, the eukaryotic glycoprotein secretion checkpoint.
Roversi, P., Marti, L., Caputo, A.T., Alonzi, D.S., Hill, J.C., Dent, K.C., Kumar, A., Levasseur, M.D., Lia, A., Waksman, T., Basu, S., Soto Albrecht, Y., Qian, K., McIvor, J.P., Lipp, C.B., Siliqi, D., Vasiljevic, S., Mohammed, S., Lukacik, P., Walsh, M.A., Santino, A., Zitzmann, N.(2017) Proc Natl Acad Sci U S A 114: 8544-8549
- PubMed: 28739903
- DOI: https://doi.org/10.1073/pnas.1703682114
- Primary Citation Related Structures:
5MU1, 5MZO, 5N2J, 5NV4 - PubMed Abstract:
Glycoproteins traversing the eukaryotic secretory pathway begin life in the endoplasmic reticulum (ER), where their folding is surveyed by the 170-kDa UDP-glucose:glycoprotein glucosyltransferase (UGGT). The enzyme acts as the single glycoprotein folding quality control checkpoint: it selectively reglucosylates misfolded glycoproteins, promotes their association with ER lectins and associated chaperones, and prevents premature secretion from the ER. UGGT has long resisted structural determination and sequence-based domain boundary prediction. Questions remain on how this single enzyme can flag misfolded glycoproteins of different sizes and shapes for ER retention and how it can span variable distances between the site of misfold and a glucose-accepting N-linked glycan on the same glycoprotein. Here, crystal structures of a full-length eukaryotic UGGT reveal four thioredoxin-like (TRXL) domains arranged in a long arc that terminates in two β-sandwiches tightly clasping the glucosyltransferase domain. The fold of the molecule is topologically complex, with the first β-sandwich and the fourth TRXL domain being encoded by nonconsecutive stretches of sequence. In addition to the crystal structures, a 15-Å cryo-EM reconstruction reveals interdomain flexibility of the TRXL domains. Double cysteine point mutants that engineer extra interdomain disulfide bridges rigidify the UGGT structure and exhibit impaired activity. The intrinsic flexibility of the TRXL domains of UGGT may therefore endow the enzyme with the promiscuity needed to recognize and reglucosylate its many different substrates and/or enable reglucosylation of N-linked glycans situated at variable distances from the site of misfold.
- Oxford Glycobiology Institute, Department of Biochemistry, University of Oxford, Oxford OX1 3QU, United Kingdom; pietro.roversi@bioch.ox.ac.uk nicole.zitzmann@bioch.ox.ac.uk.
Organizational Affiliation: