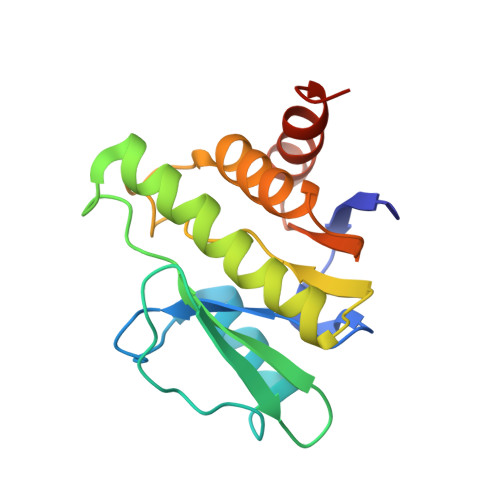
Conformational plasticity of the VEEV macro domain is important for binding of ADP-ribose.
Makrynitsa, G.I., Ntonti, D., Marousis, K.D., Birkou, M., Matsoukas, M.T., Asami, S., Bentrop, D., Papageorgiou, N., Canard, B., Coutard, B., Spyroulias, G.A.(2019) J Struct Biol 206: 119-127
- PubMed: 30825649 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jsb.2019.02.008
- Primary Citation Related Structures:
5ISN, 5MQX - PubMed Abstract:
Venezuelan equine encephalitis virus (VEEV) is a new world alphavirus which can be involved in several central nervous system disorders such as encephalitis and meningitis. The VEEV genome codes for 4 non-structural proteins (nsP), of which nsP3 contains a Macro domain. Macro domains (MD) can be found as stand-alone proteins or embedded within larger proteins in viruses, bacteria and eukaryotes. Their most common feature is the binding of ADP-ribose (ADPr), while several macro domains act as ribosylation writers, erasers or readers. Alphavirus MD erase ribosylation but their precise contribution in viral replication is still under investigation. NMR-driven titration experiments of ADPr in solution with the VEEV macro domain (in apo- and complex state) show that it adopts a suitable conformation for ADPr binding. Specific experiments indicate that the flexibility of the loops β5-α3 and α3-β6 is critical for formation of the complex and assists a wrapping mechanism for ADPr binding. Furthermore, along with this sequence of events, the VEEV MD undergoes a conformational exchange process between the apo state and a low-populated "dark" conformational state.
- Department of Pharmacy, University of Patras, GR-26504, Greece.
Organizational Affiliation: