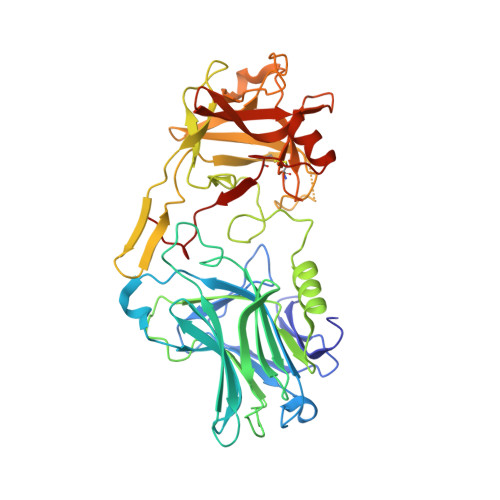
Crystal structure of the BoNT/A2 receptor-binding domain in complex with the luminal domain of its neuronal receptor SV2C.
Benoit, R.M., Scharer, M.A., Wieser, M.M., Li, X., Frey, D., Kammerer, R.A.(2017) Sci Rep 7: 43588-43588
- PubMed: 28252640
- DOI: https://doi.org/10.1038/srep43588
- Primary Citation Related Structures:
5MOY - PubMed Abstract:
A detailed molecular understanding of botulinum neurotoxin (BoNT)/host-cell-receptor interactions is fundamental both for developing strategies against botulism and for generating improved BoNT variants for medical applications. The X-ray crystal structure of the receptor-binding domain (H C ) of BoNT/A1 in complex with the luminal domain (LD) of its neuronal receptor SV2C revealed only few specific side-chain - side-chain interactions that are important for binding. Notably, two BoNT/A1 residues, Arg 1156 and Arg 1294, that are crucial for the interaction with SV2, are not conserved among subtypes. Because it has been suggested that differential receptor binding of subtypes might explain their differences in biological activity, we determined the crystal structure of BoNT/A2-H C in complex with SV2C-LD. Although only few side-chain interactions are conserved between the two BoNT/A subtypes, the overall binding mode of subtypes A1 and A2 is virtually identical. In the BoNT/A2-H C - SV2C complex structure, a missing cation-π stacking is compensated for by an additional salt bridge and an anion-π stacking interaction, which explains why the binding of BoNT/A subtypes to SV2C tolerates variable side chains. These findings suggest that motif extensions and a shallow binding cleft in BoNT/A-H C contribute to binding specificity.
- Laboratory of Biomolecular Research, Division of Biology and Chemistry, Paul Scherrer Institute, CH-5232 Villigen PSI, Switzerland.
Organizational Affiliation: