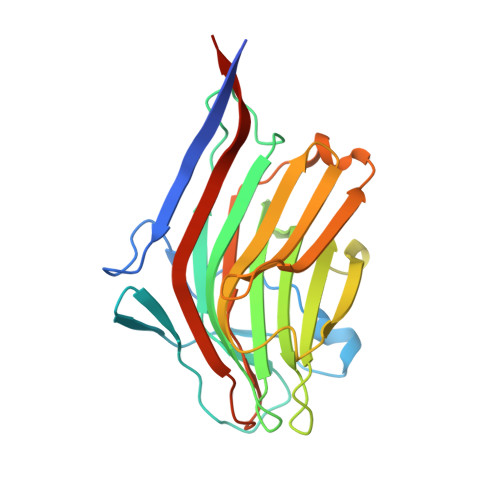
Evolved distal tail carbohydrate binding modules of Lactobacillus phage J-1: a novel type of anti-receptor widespread among lactic acid bacteria phages.
Dieterle, M.E., Spinelli, S., Sadovskaya, I., Piuri, M., Cambillau, C.(2017) Mol Microbiol 104: 608-620
- PubMed: 28196397 Search on PubMed
- DOI: https://doi.org/10.1111/mmi.13649
- Primary Citation Related Structures:
5LY8 - PubMed Abstract:
Bacteriophage replication requires specific host-recognition. Some siphophages harbour a large complex, the baseplate, at the tip of their non-contractile tail. This baseplate holds receptor binding proteins (RBPs) that can recognize the host cell-wall polysaccharide (CWPS) and specifically attach the phage to its host. While most phages possess a dedicated RBP, the phage J-1 that infects Lactobacillus casei seemed to lack one. It has been shown that the phage J-1 distal tail protein (Dit) plays a role in host recognition and that its sequence comprises two inserted modules compared with 'classical' Dits. The first insertion is similar to carbohydrate-binding modules (CBMs), whereas the second insertion remains undocumented. Here, we determined the structure of the second insertion and found it also similar to several CBMs. Expressed insertion CBM2, but not CBM1, binds to L. casei cells and neutralize phage attachment to the bacterial cell wall and the isolated and purified CWPS of L. casei BL23 prevents CBM2 attachment to the host. Electron microscopy single particle reconstruction of the J-1 virion baseplate revealed that CBM2 is projected at the periphery of Dit to optimally bind the CWPS receptor. Taken together, these results identify J-1 evolved Dit as the phage RBP.
- Departamento de Química Biológica, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires, IQUIBICEN-CONICET, Buenos Aires, Argentina.
Organizational Affiliation: