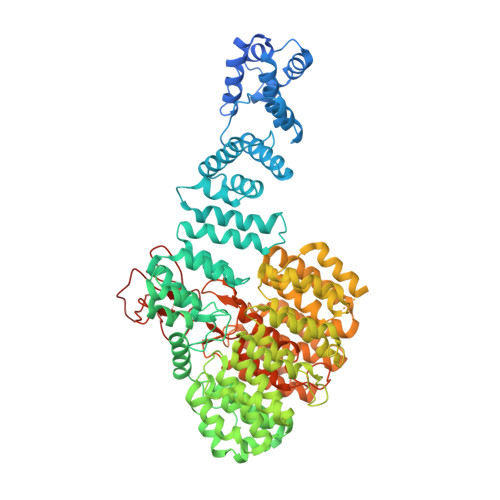
Molecular Details Underlying Dynamic Structures and Regulation of the Human 26S Proteasome.
Wang, X., Cimermancic, P., Yu, C., Schweitzer, A., Chopra, N., Engel, J.L., Greenberg, C., Huszagh, A.S., Beck, F., Sakata, E., Yang, Y., Novitsky, E.J., Leitner, A., Nanni, P., Kahraman, A., Guo, X., Dixon, J.E., Rychnovsky, S.D., Aebersold, R., Baumeister, W., Sali, A., Huang, L.(2017) Mol Cell Proteomics 16: 840-854
- PubMed: 28292943
- DOI: https://doi.org/10.1074/mcp.M116.065326
- Primary Citation of Related Structures:
5LN3 - PubMed Abstract:
The 26S proteasome is the macromolecular machine responsible for ATP/ubiquitin dependent degradation. As aberration in proteasomal degradation has been implicated in many human diseases, structural analysis of the human 26S proteasome complex is essential to advance our understanding of its action and regulation mechanisms. In recent years, cross-linking mass spectrometry (XL-MS) has emerged as a powerful tool for elucidating structural topologies of large protein assemblies, with its unique capability of studying protein complexes in cells. To facilitate the identification of cross-linked peptides, we have previously developed a robust amine reactive sulfoxide-containing MS-cleavable cross-linker, disuccinimidyl sulfoxide (DSSO). To better understand the structure and regulation of the human 26S proteasome, we have established new DSSO-based in vivo and in vitro XL-MS workflows by coupling with HB-tag based affinity purification to comprehensively examine protein-protein interactions within the 26S proteasome. In total, we have identified 447 unique lysine-to-lysine linkages delineating 67 interprotein and 26 intraprotein interactions, representing the largest cross-link dataset for proteasome complexes. In combination with EM maps and computational modeling, the architecture of the 26S proteasome was determined to infer its structural dynamics. In particular, three proteasome subunits Rpn1, Rpn6, and Rpt6 displayed multiple conformations that have not been previously reported. Additionally, cross-links between proteasome subunits and 15 proteasome interacting proteins including 9 known and 6 novel ones have been determined to demonstrate their physical interactions at the amino acid level. Our results have provided new insights on the dynamics of the 26S human proteasome and the methodologies presented here can be applied to study other protein complexes.
- From the ‡Department of Physiology & Biophysics, University of California, Irvine, Irvine, California 92697.
Organizational Affiliation: