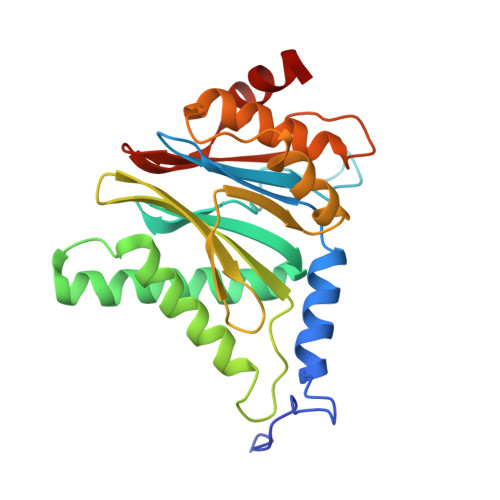
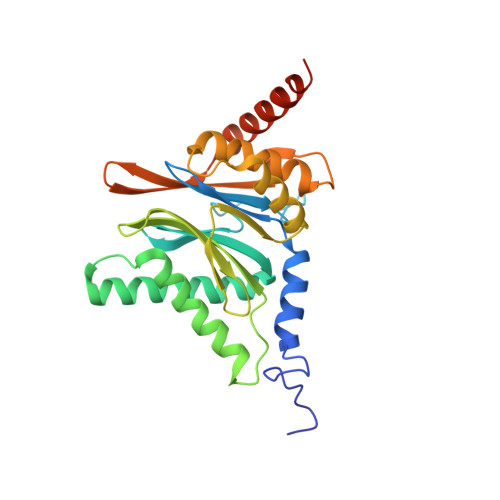
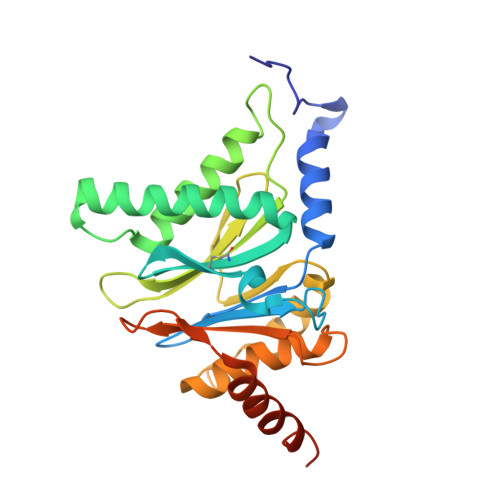
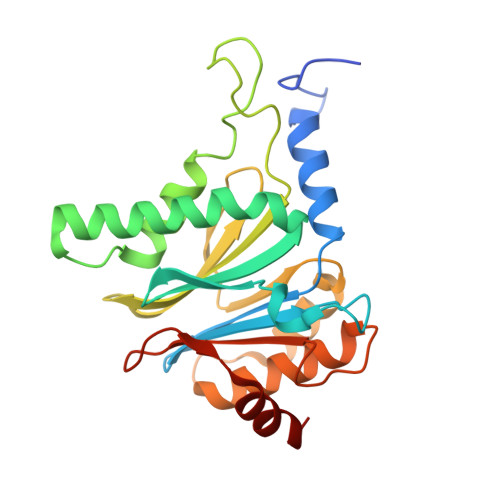
The inhibition mechanism of human 20S proteasomes enables next-generation inhibitor design.
Schrader, J., Henneberg, F., Mata, R.A., Tittmann, K., Schneider, T.R., Stark, H., Bourenkov, G., Chari, A.(2016) Science 353: 594-598
- PubMed: 27493187 Search on PubMed
- DOI: https://doi.org/10.1126/science.aaf8993
- Primary Citation Related Structures:
5LE5, 5LEX, 5LEY, 5LEZ, 5LF0, 5LF1, 5LF3, 5LF4, 5LF6, 5LF7 - PubMed Abstract:
The proteasome is a validated target for anticancer therapy, and proteasome inhibition is employed in the clinic for the treatment of tumors and hematological malignancies. Here, we describe crystal structures of the native human 20S proteasome and its complexes with inhibitors, which either are drugs approved for cancer treatment or are in clinical trials. The structure of the native human 20S proteasome was determined at an unprecedented resolution of 1.8 angstroms. Additionally, six inhibitor-proteasome complex structures were elucidated at resolutions between 1.9 and 2.1 angstroms. Collectively, the high-resolution structures provide new insights into the catalytic mechanisms of inhibition and necessitate a revised description of the proteasome active site. Knowledge about inhibition mechanisms provides insights into peptide hydrolysis and can guide strategies for the development of next-generation proteasome-based cancer therapeutics.
- Department of Structural Dynamics, Max Planck Institut für biophysikalische Chemie, Am Fassberg 11, D-37077 Göttingen, Germany.
Organizational Affiliation: