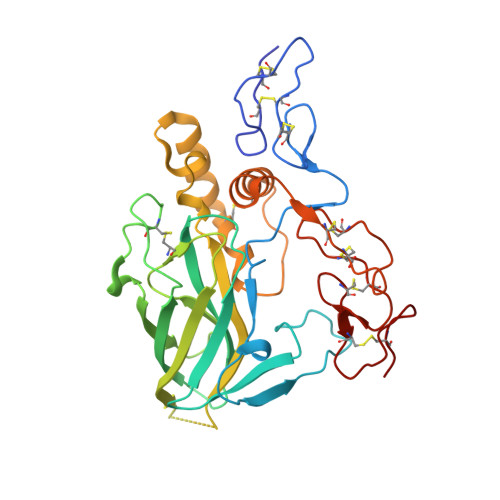
Crystallographic analysis of the laminin beta 2 short arm reveals how the LF domain is inserted into a regular array of LE domains.
Pulido, D., Briggs, D.C., Hua, J., Hohenester, E.(2017) Matrix Biol 57-58: 204-212
- PubMed: 27425256 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.matbio.2016.06.006
- Primary Citation Related Structures:
5LF2 - PubMed Abstract:
Laminins are a major constituent of all basement membranes. The polymerisation of laminins at the cell surface is mediated by the three short arms of the cross-shaped laminin heterotrimer. The short arms contain repeats of laminin-type epidermal growth factor-like (LE) domains, interspersed with globular domains of unknown function. A single LF domain is inserted between LE5 and LE6 of the laminin β1 and β2 chains. We report the crystal structure at 1.85Å resolution of the laminin β2 LE5-LF-LE6 region. The LF domain consists of a β-sandwich related to bacterial family 35 carbohydrate binding modules, and more distantly to the L4 domains present in the short arms of laminin α and γ chains. An α-helical region mediates the extensive interaction of the LF domain with LE5. The relative arrangement of LE5 and LE6 is very similar to that of consecutive LE domains in uninterrupted LE tandems. Fitting atomic models to a low-resolution structure of the first eight domains of the laminin β1 chain determined by small-angle X-ray scattering suggests a deviation from the regular LE array at the LE4-LE5 junction. These results advance our understanding of laminin structure.
- Department of Life Sciences, Imperial College London, UK.
Organizational Affiliation: