Structural Biology for Drug Design: Applications in Two Systems
Maize, K.(2016) Thesis
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Histidine triad nucleotide-binding protein 1 | 129 | Homo sapiens | Mutation(s): 0 Gene Names: HINT1, HINT EC: 3 (PDB Primary Data), 3.4.22 (UniProt), 3.9.1 (UniProt) | 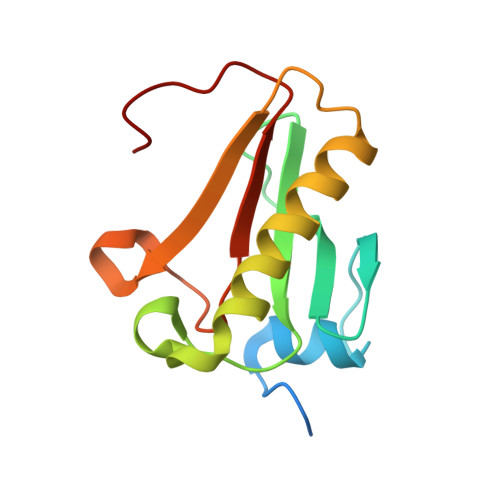 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P49773 GTEx: ENSG00000169567 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P49773 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 6UU Download:Ideal Coordinates CCD File | C [auth A] | [2-(1~{H}-indol-3-yl)ethylamino]phosphonic acid C10 H13 N2 O3 P LZBMOXGGHSGOHA-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 78.093 | α = 90 |
| b = 46.151 | β = 94.66 |
| c = 63.994 | γ = 90 |
| Software Name | Purpose |
|---|---|
| Aimless | data scaling |
| PHASER | phasing |
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |