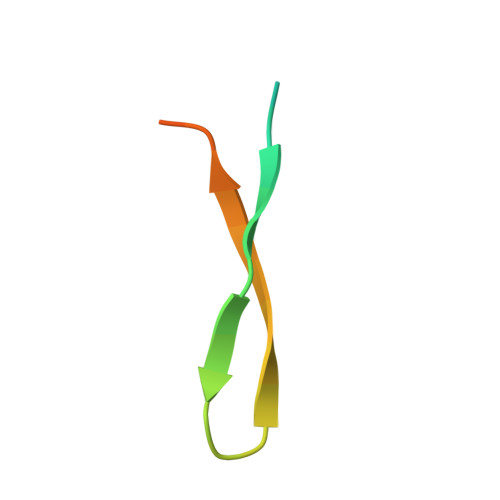
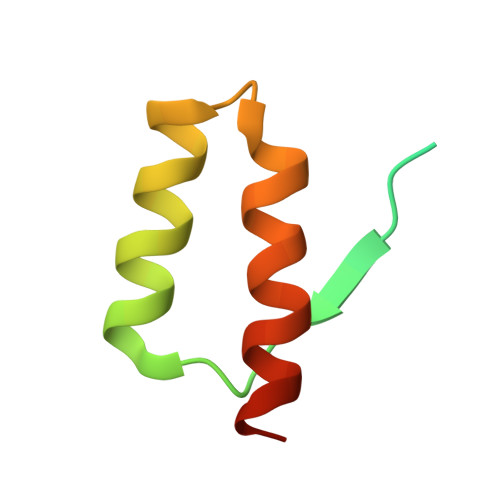
beta-Hairpin of Islet Amyloid Polypeptide Bound to an Aggregation Inhibitor.
Mirecka, E.A., Feuerstein, S., Gremer, L., Schroder, G.F., Stoldt, M., Willbold, D., Hoyer, W.(2016) Sci Rep 6: 33474-33474
- PubMed: 27641459 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/srep33474
- Primary Citation Related Structures:
5K5G - PubMed Abstract:
In type 2 diabetes, the formation of islet amyloid consisting of islet amyloid polypeptide (IAPP) is associated with reduction in β-cell mass and contributes to the failure of islet cell transplantation. Rational design of inhibitors of IAPP amyloid formation has therapeutic potential, but is hampered by the lack of structural information on inhibitor complexes of the conformationally flexible, aggregation-prone IAPP. Here we characterize a β-hairpin conformation of IAPP in complex with the engineered binding protein β-wrapin HI18. The β-strands correspond to two amyloidogenic motifs, 12-LANFLVH-18 and 22-NFGAILS-28, which are connected by a turn established around Ser-20. Besides backbone hydrogen bonding, the IAPP:HI18 interaction surface is dominated by non-polar contacts involving hydrophobic side chains of the IAPP β-strands. Apart from monomers, HI18 binds oligomers and fibrils and inhibits IAPP aggregation and toxicity at low substoichiometric concentrations. The IAPP β-hairpin can serve as a molecular recognition motif enabling control of IAPP aggregation.
- Institute of Physical Biology, Heinrich Heine University Düsseldorf, 40204 Düsseldorf, Germany.
Organizational Affiliation: