The crystal structure of CDYL2 domain of human CDYL2 protein
WU, H., DONG, A., ZENG, H., ELBAKKOURI, M., BARSYTE, D., VEDADI, M., TATLOCK, J., OWEN, D., BUNNAGE, M., Bountra, C., Arrowsmith, C.H., Edwards, A.M., BROWN, P.J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Chromodomain Y-like protein 2 | 83 | Homo sapiens | Mutation(s): 0 Gene Names: CDYL2 | 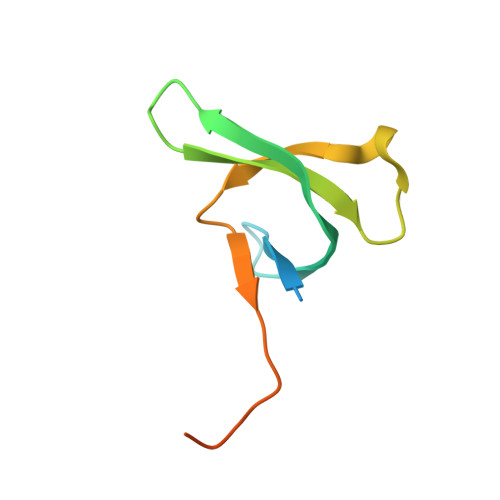 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q8N8U2 GTEx: ENSG00000166446 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q8N8U2 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| LYS-LYS-LYS-ALA-ARG-MLY-SER-ALA-GLY-ALA-ALA-LYS-TYR | 13 | Homo sapiens | Mutation(s): 0 | 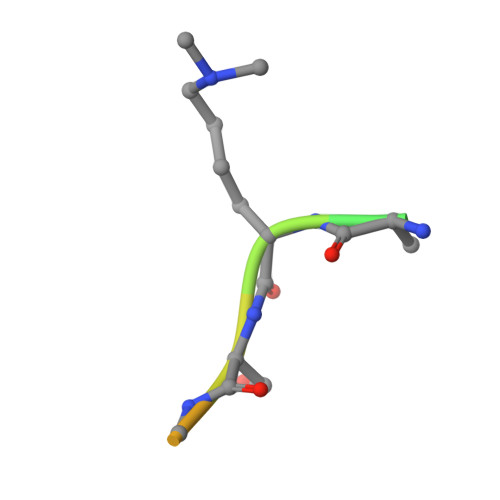 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P10412 GTEx: ENSG00000168298 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P10412 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MLY Query on MLY | B | L-PEPTIDE LINKING | C8 H18 N2 O2 |  | LYS |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 57.962 | α = 90 |
| b = 57.962 | β = 90 |
| c = 98.824 | γ = 120 |
| Software Name | Purpose |
|---|---|
| SCALEPACK | data scaling |
| MOLREP | phasing |
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |
| HKL-3000 | data reduction |