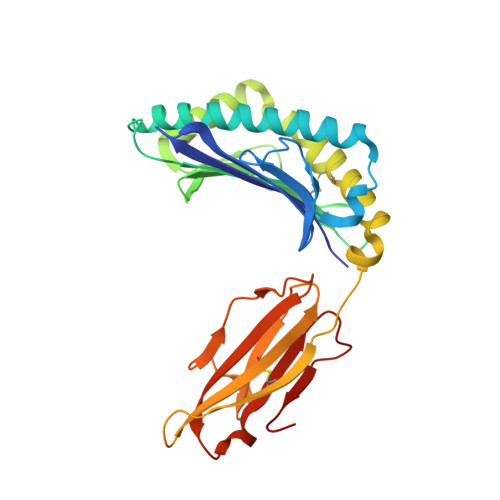
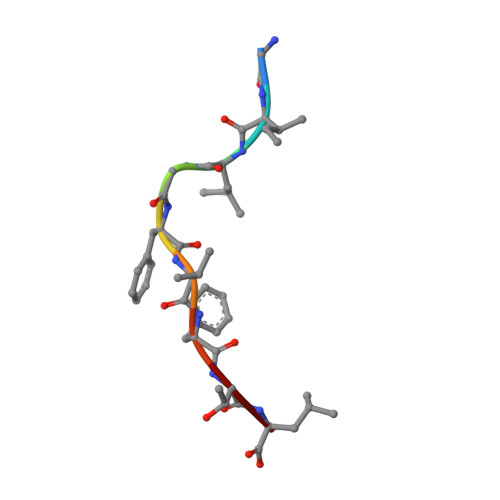
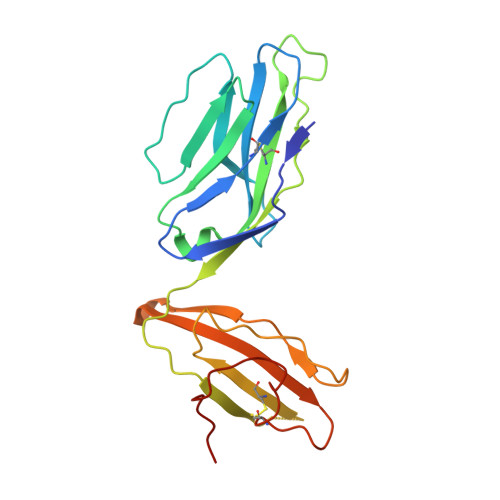
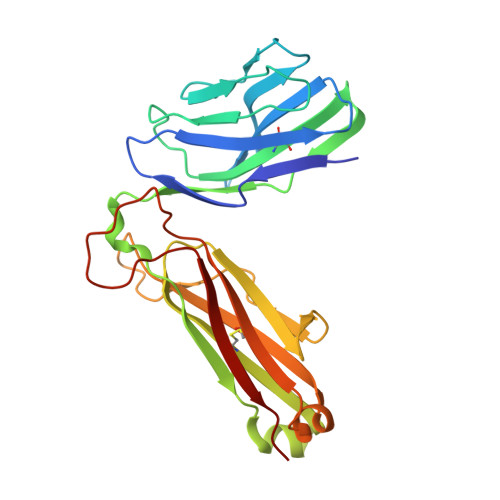
Broad TCR repertoire and diverse structural solutions for recognition of an immunodominant CD8(+) T cell epitope.
Song, I., Gil, A., Mishra, R., Ghersi, D., Selin, L.K., Stern, L.J.(2017) Nat Struct Mol Biol 24: 395-406
- PubMed: 28250417 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nsmb.3383
- Primary Citation Related Structures:
5ISZ, 5JHD - PubMed Abstract:
A keystone of antiviral immunity is CD8 + T cell recognition of viral peptides bound to MHC-I proteins. The recognition modes of individual T cell receptors (TCRs) have been studied in some detail, but the role of TCR variation in providing a robust response to viral antigens is unclear. The influenza M1 epitope is an immunodominant target of CD8 + T cells that help to control influenza in HLA-A2 + individuals. Here we show that CD8 + T cells use many distinct TCRs to recognize HLA-A2-M1, which enables the use of different structural solutions to the problem of specifically recognizing a relatively featureless peptide antigen. The vast majority of responding TCRs target a small cleft between HLA-A2 and the bound M1 peptide. These broad repertoires lead to plasticity in antigen recognition and protection against T cell clonal loss and viral escape.
- Department of Pathology, University of Massachusetts Medical School, Worcester, Massachusetts, USA.
Organizational Affiliation: