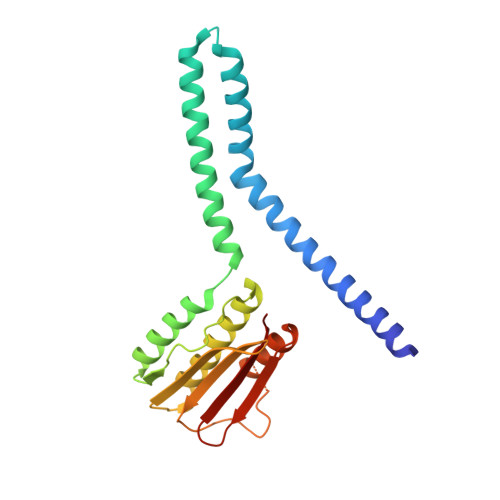
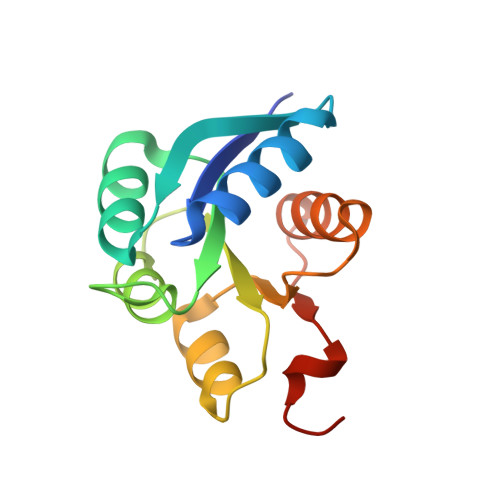
Regulation of signaling directionality revealed by 3D snapshots of a kinase:regulator complex in action.
Trajtenberg, F., Imelio, J.A., Machado, M.R., Larrieux, N., Marti, M.A., Obal, G., Mechaly, A.E., Buschiazzo, A.(2016) Elife 5
- PubMed: 27938660 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.7554/eLife.21422
- Primary Citation Related Structures:
5IUJ, 5IUK, 5IUL, 5IUM - PubMed Abstract:
Two-component systems (TCS) are protein machineries that enable cells to respond to input signals. Histidine kinases (HK) are the sensory component, transferring information toward downstream response regulators (RR). HKs transfer phosphoryl groups to their specific RRs, but also dephosphorylate them, overall ensuring proper signaling. The mechanisms by which HKs discriminate between such disparate directions, are yet unknown. We now disclose crystal structures of the HK:RR complex DesK:DesR from Bacillus subtilis , comprising snapshots of the phosphotransfer and the dephosphorylation reactions. The HK dictates the reactional outcome through conformational rearrangements that include the reactive histidine. The phosphotransfer center is asymmetric, poised for dissociative nucleophilic substitution. The structural bases of HK phosphatase/phosphotransferase control are uncovered, and the unexpected discovery of a dissociative reactional center, sheds light on the evolution of TCS phosphotransfer reversibility. Our findings should be applicable to a broad range of signaling systems and instrumental in synthetic TCS rewiring.
- Laboratory of Molecular and Structural Microbiology, Institut Pasteur de Montevideo, Montevideo, Uruguay.
Organizational Affiliation: