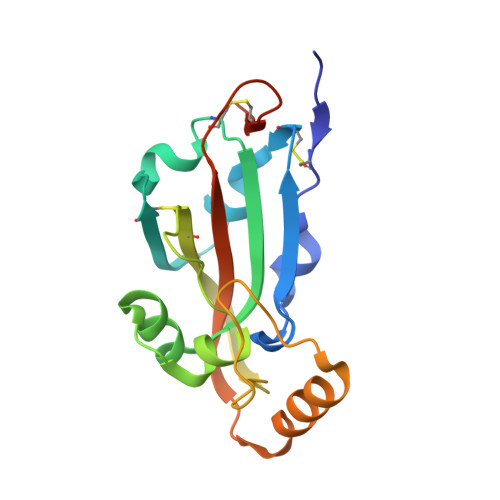
Putative Receptor Binding Domain of Bat-Derived Coronavirus HKU9 Spike Protein: Evolution of Betacoronavirus Receptor Binding Motifs
Huang, C., Qi, J., Lu, G., Wang, Q., Yuan, Y., Wu, Y., Zhang, Y., Yan, J., Gao, G.F.(2016) Biochemistry 55: 5977-5988
- PubMed: 27696819 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/acs.biochem.6b00790
- Primary Citation Related Structures:
5GYQ - PubMed Abstract:
The suggested bat origin for Middle East respiratory syndrome coronavirus (MERS-CoV) has revitalized the studies of other bat-derived coronaviruses with respect to interspecies transmission potential. Bat coronavirus (BatCoV) HKU9 is an important betacoronavirus (betaCoV) that is phylogenetically affiliated with the same genus as MERS-CoV. The bat surveillance data indicated that BatCoV HKU9 has been widely spreading and circulating in bats. This highlights the necessity of characterizing the virus for its potential to cross species barriers. The receptor binding domain (RBD) of the coronavirus spike (S) protein recognizes host receptors to mediate virus entry and is therefore a key factor determining the viral tropism and transmission capacity. In this study, the putative S RBD of BatCoV HKU9 (HKU9-RBD), which is homologous to other betaCoV RBDs that have been structurally and functionally defined, was characterized via a series of biophysical and crystallographic methods. By using surface plasmon resonance, we demonstrated that HKU9-RBD binds to neither SARS-CoV receptor ACE2 nor MERS-CoV receptor CD26. We further determined the atomic structure of HKU9-RBD, which as expected is composed of a core and an external subdomain. The core subdomain fold resembles those of other betaCoV RBDs, whereas the external subdomain is structurally unique with a single helix, explaining the inability of HKU9-RBD to react with either ACE2 or CD26. Via comparison of the available RBD structures, we further proposed a homologous intersubdomain binding mode in betaCoV RBDs that anchors the external subdomain to the core subdomain. The revealed RBD features would shed light on the evolution route of betaCoV.
- National Institute for Viral Disease Control and Prevention, Chinese Center for Disease Control and Prevention (China CDC) , Beijing 102206, China.
Organizational Affiliation: